3-Hydroxybenzaldehyde
Chemical compound
From Wikipedia, the free encyclopedia
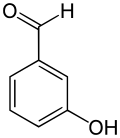
3-Hydroxybenzaldehyde is an organic compound with the formula HOC6H4CHO. It is a colorless solid although most samples appear tan. Two other isomers of hydroxybenzaldehyde exist.

 | |
| Names | |
|---|---|
| Preferred IUPAC name
3-Hydroxybenzaldehyde | |
| Other names
m-Hydroxybenzaldehyde; m-Formylphenol; 3-formylphenol[1] | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.630 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C7H6O2 | |
| Molar mass | 122.123 g·mol−1 |
| Appearance | colorless solid |
| Density | 1.1179 g/cm3 (130 °C)[1] |
| Melting point | 106 °C (223 °F; 379 K)[1] |
| Boiling point | 240 °C (464 °F; 513 K)[1] |
| Acidity (pKa) | 8.98 (25 °C)[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Preparation
It has been prepared from 3-nitrobenzaldehyde in a sequence of nitro group reduction, diazotization of the amine, and hydrolysis.[3][4]
3-hydroxybenzyl-alcohol dehydrogenase is an nicotinamide adenine dinucleotide phosphate-dependent enzyme that produces 3-hydroxybenzaldehyde from 3-hydroxybenzyl alcohol.[5][6]
Biomedical properties
See also
- Salicylaldehyde (2-hydroxybenzaldehyde)
- 4-Hydroxybenzaldehyde

