Ditolylguanidine
Experimental drug
From Wikipedia, the free encyclopedia
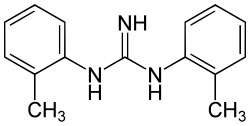
Ditolylguanidine (DTG) is a sigma receptor agonist.[1] It is somewhat[2] selective for sigma receptors, but non-selective between the two sigma receptor subtypes, binding to both σ1 and σ2 with equal affinity.[3] It has neuroprotective[4] and antidepressant effects,[5] and potentiates the effects of NMDA antagonists.[6]

 | |
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.002.344 |
| Chemical and physical data | |
| Formula | C15H17N3 |
| Molar mass | 239.322 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Properties
N,N′-Di-o-tolylguanidine is a flammable, white solid that is practically insoluble in water. It decomposes upon heating, potentially releasing nitrogen oxides, carbon monoxide, and carbon dioxide.[7]
Safety Information
N,N′-Di-o-tolylguanidine is not itself carcinogenic, but due to the manufacturing process, it may still contain o-toluidine, which is carcinogenic. Therefore, if the o-toluidine content exceeds 0.1 percent, the mixture should be considered carcinogenic.[7]
