Combretastatin A-4
Chemical compound
From Wikipedia, the free encyclopedia
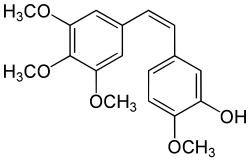
Combretastatin A-4 is a combretastatin and a stilbenoid. It can be isolated from Combretum afrum, the Eastern Cape South African bushwillow tree or in Combretum leprosum, the mofumbo, a species found in Brazil.[3][4]

 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methoxy-5-[(1Z)-2-(3,4,5-trimethoxyphenyl)ethen-1-yl]phenol | |
| Other names
Combretastatin A4 CA-4 1-(3,4,5-Trimethoxyphenyl)-2-(3′-hydroxy-4′-methoxyphenyl)ethene 3,4,5-Trimethoxy-3′-hydroxy-4′-methoxystilbene | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.159.667 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C18H20O5 | |
| Molar mass | 316.34 g/mol |
| Melting point | 116 °C (241 °F; 389 K)[1] |
| insoluble | |
| Solubility in DMSO, Ethanol | DMSO : 63 mg/mL, Ethanol : 34 mg/mL[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Function
Tubulin represents a potent target in cancer chemotherapy, given its role in cell division. Combretastatin is a naturally occurring well known tubulin polymerization inhibitor. Combretastatin A-4 comes in two stereoisomers (cis (shown top right), and trans); The cis form binds much better to the 'colchicine' site on tubulin to inhibit polymerization.[5]
Derivatives
Combretastatin A-4 is the active component of combretastatin A-4 phosphate, a prodrug designed to damage the vasculature (blood vessels) of cancer tumors causing central necrosis.[citation needed]
A large number of synthetic derivatives have been reported,[6][7] including beta-lactam based compounds.[8]
Pharmacokinetics
CA4 has a half life of 1.8-4.2h in humans. CA4P(a prodrug) has a half life of 0.22-0.36h in humans.[9]
See also
- Ombrabulin, a combretastatin A-4 derivative in clinical trials for treatment of cancer
