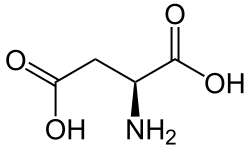
Iminosuccinic acid
Chemical compound
From Wikipedia, the free encyclopedia
Iminoaspartic acid (also known as iminosuccinate or iminoaspartate) is a dicarboxylic acid used in the biosynthesis of nicotinic acid and nicotinamide adenine dinucleotide.

 | |
| Names | |
|---|---|
| IUPAC name
2-Iminobutanedioic acid | |
| Other names
Iminoaspartic acid; 2-iminobutanedioic acid, iminosuccinate, alpha-iminosuccinate, iminosuccinic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H5NO4 | |
| Molar mass | 131.087 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Biosynthesis
The enzyme L-aspartate oxidase oxidises L-aspartic acid:[1]
Subsequent conversions
In Escherichia coli another enzyme, quinolinate synthase, takes iminosuccinic acid with dihydroxyacetone phosphate to form quinolinic acid.[2]
This iron-sulfur protein requires a [4Fe-4S] cluster for activity. The quinolinic acid can be converted to nicotinic acid or incorporated into nicotinamide adenine dinucleotide.[3]