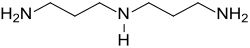
Norspermidine
Chemical compound
From Wikipedia, the free encyclopedia
Norspermidine is a polyamine of similar structure to the more common spermidine. Norspermidine has been found to occur naturally in some species of plants,[1][2] bacteria,[3] and algae.[4]

 | |
| Names | |
|---|---|
| Preferred IUPAC name
N1-(3-Aminopropyl)propane-1,3-diamine | |
| Identifiers | |
3D model (JSmol) |
|
| 1071254 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.000.238 |
| EC Number |
|
| 26839 | |
| KEGG | |
| MeSH | norspermidine |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2269 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H17N3 | |
| Molar mass | 131.223 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Ichtyal, ammoniacal |
| Density | 938 mg mL−1 |
| Melting point | −16 to 0 °C; 3 to 32 °F; 257 to 273 K |
| Boiling point | 240.60 °C; 465.08 °F; 513.75 K |
| log P | −0.826 |
Refractive index (nD) |
1.481–1.482 |
| Hazards | |
| GHS labelling: | |
  | |
| Danger | |
| H302, H311, H314, H317, H330 | |
| P260, P280, P284, P305+P351+P338, P310 | |
| Flash point | 117 °C (243 °F; 390 K) |
| 280 °C (536 °F; 553 K) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
738 mg kg−1 (oral, rat) |
| Safety data sheet (SDS) | fishersci.com |
| Related compounds | |
Related amines |
|
Related compounds |
Agmatine |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Norspermidine is being researched for use as a cancer medication.[5][6]
Biosynthesis
Norspermidine is an aliphatic polyamine. In ϵ-proteobacteria, which are found in human gut microbiota, a combination of two enzymes is used to produce norspermidine from 1,3-diaminopropane.[7] First, carboxynorspermidine synthase catalyses a reductive amination using nicotinamide adenine dinucleotide phosphate (NADPH) as the reducing agent.[8][9]
The intermediate, carboxynorspermidine, is then decarboxylated by carboxynorspermidine decarboxylase:[7][9]