Nitrosylsulfuric acid
Chemical compound
From Wikipedia, the free encyclopedia
Nitrosylsulfuric acid is the chemical compound with the formula NOHSO4. It is a colourless solid that is used industrially in the production of caprolactam,[3] and was formerly part of the lead chamber process for producing sulfuric acid. The compound is a salt best described as NO+HSO4− (nitrosonium hydrogensulfate).[4] It is called “lead-chamber crystals”.
 | |
| Names | |
|---|---|
| IUPAC name
Nitrosylsulfuric acid | |
| Other names
nitrosonium bisulfate, chamber crystals | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.029.058 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| NOHSO4 | |
| Molar mass | 127.08 g/mol |
| Appearance | Pale yellow crystals[1] |
| Density | 1.865 g/mL in 40% sulfuric acid soln [2] |
| Melting point | 70 °C (158 °F; 343 K)[1] |
| Boiling point | Decomposes |
| Decomposes | |
| Solubility | Soluble in H2SO4[1] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Oxidizer |
| Related compounds | |
Other anions |
NOCl |
Other cations |
NaHSO4 |
Related compounds |
NOBF4 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
In organic chemistry, it is used as a reagent for nitrosating, as a diazotizing agent, and as an oxidizing agent.[1]
Synthesis and reactions
A typical procedure entails dissolving sodium nitrite in cold sulfuric acid:[5][6]
- HNO2 + H2SO4 → NOHSO4 + H2O
It can also be prepared by the reaction of nitric acid and sulfur dioxide.[7]
NOHSO4 is used in organic chemistry to prepare diazonium salts from amines,[5][6] for example in the Sandmeyer reaction. Related NO-delivery reagents include nitrosonium tetrafluoroborate [NO]+[BF4]− and nitrosyl chloride.
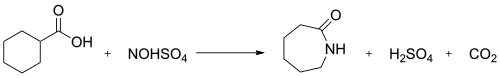
In industry, the nitrosodecarboxylation reaction between nitrosylsulfuric acid and cyclohexanecarboxylic acid is used to generate caprolactam:[3] This conversion illustrates the Snia Viscosa process