Knoevenagel condensation
Organic chemical reaction
From Wikipedia, the free encyclopedia
In organic chemistry, the Knoevenagel condensation (pronounced [ˈknøːvənaːɡl̩]) reaction is a type of chemical reaction named after German chemist Emil Knoevenagel. It is a modification of the aldol condensation.[1][2]
| Knoevenagel condensation | |
|---|---|
| Named after | Emil Knoevenagel |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | knoevenagel-condensation |
| RSC ontology ID | RXNO:0000044 |
A Knoevenagel condensation is a nucleophilic addition of an active hydrogen compound to a carbonyl group followed by a dehydration reaction in which a molecule of water is eliminated (hence condensation). The product is often an α,β-unsaturated ketone (a conjugated enone).

In this reaction the carbonyl group is an aldehyde or a ketone. The catalyst is usually a weakly basic amine. The active hydrogen component has the forms:[3]
- Z−CH2−Z or Z−CHR−Z for instance diethyl malonate, Meldrum's acid, ethyl acetoacetate or malonic acid, or cyanoacetic acid.[1]
- Z−CHRR', for instance nitromethane.
where Z is an electron withdrawing group. Z must be powerful enough to facilitate deprotonation to the enolate ion even with a mild base. Using a strong base in this reaction would induce self-condensation of the aldehyde or ketone.
The Hantzsch pyridine synthesis, the Gewald reaction and the Feist–Benary furan synthesis all contain a Knoevenagel reaction step. The reaction also led to the discovery of CS gas.
Doebner modification

The Doebner modification of the Knoevenagel condensation entails the use of pyridine as a solvent with at least one of the withdrawing groups on the nucleophile is a carboxylic acid, for example, with malonic acid. Under these conditions the condensation is accompanied by decarboxylation.[4] For example, the reaction of acrolein and malonic acid in pyridine gives trans-2,4-entadienoic acid with one carboxylic acid group and not two.[5] Sorbic acid can be prepared similarly by replacing acrolein with crotonaldehyde.[6]
Examples and applications
A Knoevenagel condensation is demonstrated in the reaction of 2-methoxybenzaldehyde 1 with the thiobarbituric acid 2 in ethanol using piperidine as a base.[7] The resulting enone 3 is a charge transfer complex molecule.

The Knoevenagel condensation is a key step in the commercial production of the antimalarial drug lumefantrine (a component of Coartem):[8]
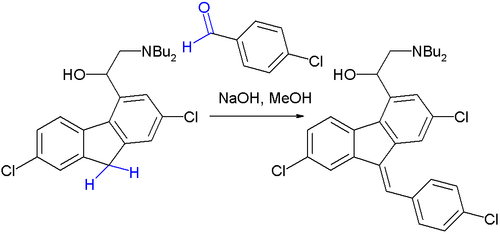
The initial reaction product is a 50:50 mixture of E and Z isomers but because both isomers equilibrate rapidly around their common hydroxyl precursor, the more stable Z-isomer can eventually be obtained.
A multicomponent reaction featuring a Knoevenagel condensation is demonstrated in this MORE synthesis with cyclohexanone, malononitrile and 3-amino-1,2,4-triazole:[9]

Weiss–Cook reaction
The Weiss–Cook reaction consists in the synthesis of cis-bicyclo[3.3.0]octane-3,7-dione employing an acetonedicarboxylic acid ester and a diacyl (1,2 ketone). The mechanism operates in the same way as the Knoevenagel condensation:[10]
