Scandium(III) phosphate
Chemical compound
From Wikipedia, the free encyclopedia
Scandium(III) phosphate is an inorganic compound of scandium, with the molecular formula ScPO4.[1]
 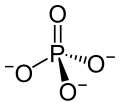 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider | |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| ScPO4 | |
| Molar mass | 139.926 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
It also occurs naturally as the rare mineral pretulite.[2][3][4]
Preparation
It can be prepared by heating scandium oxide and dilute phosphoric acid for at least a week at 400°C, yielding anhydrous scandium phosphate. Lower temperatures produce hydrates and mixtures of hydrates.[1]
Properties
Anhydrous scandium phosphate crystalizes in the zircon structure.[1]
