Iridium(IV) oxide
Chemical compound
From Wikipedia, the free encyclopedia
Iridium(IV) oxide, IrO2, is the only well-characterised oxide of iridium. It is a blue-black solid, used with other rare oxides to coat anodes.

 | |
| Names | |
|---|---|
| Other names
Iridium dioxide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.031.572 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| IrO2 | |
| Molar mass | 224.22 g/mol |
| Appearance | blue-black solid |
| Density | 11.66 g/cm3 |
| Melting point | 1,100 °C (2,010 °F; 1,370 K) decomposes |
| insoluble | |
| +224.0·10−6 cm3/mol | |
| Structure | |
| Rutile (tetragonal) | |
| Octahedral (Ir); Trigonal (O) | |
| Hazards | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions |
iridium(IV) fluoride, iridium disulfide |
Other cations |
rhodium dioxide, osmium dioxide, platinum dioxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Synthesis
As described by its discoverers, it can be formed by treating the green form of iridium trichloride with oxygen at high temperatures:
- 2 IrCl3 + 2 O2 → 2 IrO2 + 3 Cl2
A hydrated form is also known.[1]
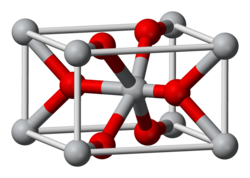
Structure
The compound adopts the TiO2 rutile structure, featuring six coordinate iridium and three coordinate oxygen.[2] It forms a tetragonal lattice with lattice parameters of 4.5Å and 3.15Å.[3]
Mechanical properties
Oxide materials are typically hard and brittle.[4] Indeed, iridium oxide does not easily deform under stress,[5] instead cracking easily.[6] Measured deflections of a thin, cantilevered iridium oxide film indicate a Young’s modulus of 300 ± 15 GPa,[5] substantially lower than the Young's modulus of metallic iridium (517 GPa).[7]
Applications
Iridium dioxide can be used to make coated electrodes[8] for industrial electrolysis or as microelectrodes for electrophysiology.[9] In electrolytic applications, IrO2 films evolve O2 efficiently.[10]
Electrode manufacture typically requires high-temperature annealing.[11]
Fracture and delamination are well-known problems when fabricating devices that incorporate iridium oxide film. One cause of delamination is lattice mismatch between iridium oxide and the substrate. Sputtering iridium oxide on a liquid crystal polymer has been proposed to avoid mismatch,[12] but sputtered films spontaneously delaminate during cyclic voltammetry if the maximum potential bias exceeds 0.9 V.[13]