Palladium(II) iodide
Chemical compound
From Wikipedia, the free encyclopedia
Palladium(II) iodide is an inorganic compound of palladium and iodine. It is commercially available, though less common than palladium(II) chloride, the usual entry point to palladium chemistry. Three polymorphs are known.[2]

 | |
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.029.276 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| I2Pd | |
| Molar mass | 360.229 g/mol |
| Appearance | Black crystals |
| Density | 6,003 g/cm3 |
| Melting point | 350 °C (decomposes) |
| Insoluble in water | |
| Hazards | |
| GHS labelling:[1] | |
 | |
| Warning | |
| H315, H319, H335 | |
| Related compounds | |
Other anions |
Palladium(II) fluoride Palladium(II) chloride Palladium(II) bromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
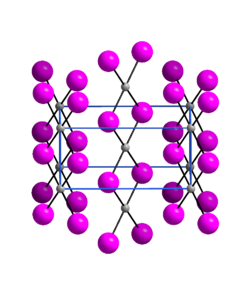
Structure
Palladium(II) iodide is an almost X-ray amorphous black powder. The α-modification has an orthorhombic crystal structure with the space group Pnmn(space group no. 58, position 5).[3]
Preparation
Palladium(II) iodide can be obtained by treating a dilute solution of palladium in nitric acid with sodium iodide at 80 °C.[2]
The high-temperature polymorph α-palladium(II) iodide can be produced by reaction of the elements at temperature above 600 °C. The γ-modification is produced as an almost amorphous powder by addition of iodide salts to aqueous H2PdCl4 solution . When heated in dilute hydrogen iodide solution, this polymorph transforms into the β phase at around 140 °C.[4]