Promethium(III) phosphate
Chemical compound
From Wikipedia, the free encyclopedia
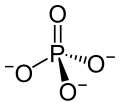
Promethium(III) phosphate is an inorganic compound, a salt of promethium and phosphate, with the chemical formula of PmPO4. It is radioactive. Its hydrate can be obtained by precipitation of soluble promethium salt and diammonium hydrogen phosphate at pH 3~4 (or obtained by hydrothermal reaction [2]), and the hydrate can be obtained by burning at 960 °C to obtain the anhydrous form.[1] Its standard enthalpy of formation is −464 kcal/mol.[3]
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| PmPO4 | |
| Molar mass | 241.89 |
| Appearance | pale yellow solid (hydrate) garnet-red solid (anhydride)[1] |
| insoluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
