Tellurium monoxide
Chemical compound
From Wikipedia, the free encyclopedia
The diatomic molecule tellurium monoxide has been found as a transient species.[2] Previous work that claimed the existence of TeO solid has not been substantiated.[3] The coating on DVDs called tellurium suboxide may be a mixture of tellurium dioxide and tellurium metal.[4]
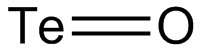 | |
| Names | |
|---|---|
| Other names
Tellurium(II) oxide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| TeO | |
| Molar mass | 143.60 g/mol |
| Related compounds | |
Other cations |
Sulfur monoxide Polonium monoxide |
| Tellurium dioxide Tellurium trioxide | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
History
Tellurium monoxide was first reported in 1883 by E. Divers and M. Shimose.[5] It was supposedly created by the thermal decomposition of tellurium sulfoxide in a vacuum,[citation needed] and was shown to react with hydrogen chloride in a 1913 report.[6] Later work has not substantiated the claim that this was a pure solid compound.[2] By 1984, the company Panasonic was working on an erasable optical disk drive containing "tellurium monoxide" (really a mixture of Te and TeO2).[7]
See also
- Tellurium dioxide
- Tellurium trioxide
- Lead carbide – originally thought to be a pure compound, but now considered more likely to be a mixture of carbon and lead
- Iodine pentabromide – originally thought to be a pure compound, but now considered to probably be a mixture of iodine monobromide and excess unreacted bromine