Thorium triiodide
Chemical compound
From Wikipedia, the free encyclopedia
Thorium triiodide is a binary inorganic compound of thorium metal and iodine with the chemical formula ThI3.[1][2][3]

 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
PubChem CID |
|
| |
| |
| Properties | |
| I3Th | |
| Molar mass | 612.7511 g·mol−1 |
| Appearance | crystals |
| reacts with water | |
| Related compounds | |
Related compounds |
Americium triiodide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Synthesis
Physical properties
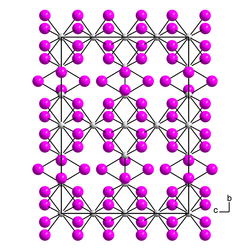
Thorium triiodide is a black, violet-tinged, usually poorly crystallized mass. Formed crystals exhibit strong dichroism from violet to olive green under the microscope and are birefringent.[5] Above 550 °C, thorium(III) iodide decomposes to thorium(IV) iodide and thorium(II) iodide. β-thorium(III) iodide has an orthorhombic crystal structure with the space group Cccm (space group no. 66).[6] The α-form has a pseudoorthorhombic crystal structure.
Chemical properties
ThI3 reacts with water to form thorium tetraiodide and thorium metal.[4]