User:Dgputt/sandbox
From Wikipedia, the free encyclopedia
- Ethylene – the simplest olefin; used as a chemical feedstock and ripening stimulant
{{Collapsible List
- Polyethylene – low-density - LDPE
- Ziplock bags
- Irrigation piping
- Gloves
- Cleanroom garments[1]
}}
- Polyethylene - linear, low-density - LLDPE
- Grocery bags
- Trash bags
- Shrink wrap
- Interior-layer for milk cartons
- Playground equipment [2]
- Polyethylene - high density - HDPE
- Water bottles
- Laundry detergent bottles
- Motor oil bottles
- Recycling bins
- Milk jugs
- Pipe
- Grocery bags
- Flower pots[3]
- Ethanol – via ethylene hydration (chemical reaction adding water)
- Acetic acid
- Vinyl acetate[4]
- Polyvinyl acetate
- Wood adhesives[5]
- Paper and packaging adhesives[6]
- Household and craft adhesives[7]
- Construction adhesives[8]
- Emulsion paints[9]
- Surface primers and sealers[10]
- Temporary protective coatings[11]
- Textile sizing agents[12]
- Paper coatings[13]
- Artistic binders and adhesives[14]
- Conservation treatments.[15]
- Pharmaceutical binders[16]
- Medical adhesives[17]
- Chewing gum base[18]
- Nonwoven fabric binders[19]
- Pressure-sensitive adhesives (copolymer systems)[20]
- Polyvinyl acetate
- Vinyl acetate[4]
- Acetic acid
- Ethylene oxide – via ethylene oxidation
- Ethylene glycol – via ethylene oxide hydration
- Engine coolant – ethylene glycol, water and inhibitor mixture
- Polyesters – any of several polymers with ester linkages in the main chain
- Glycol ethers – via glycol condescension
- Monoalkyl ethers
- Ethylene glycol monomethyl ether
- Ethylene glycol monoethyl ether
- Ethylene glycol monopropyl ether
- Ethylene glycol monoisopropyl ether
- Ethylene glycol monobutyl ether - a widely used solvent in paintings and surface coatings, cleaning products and inks
- Ethylene glycol monophenyl ether
- Ethylene glycol monobenzyl ether
- Diethylene glycol monomethyl ether
- Diethylene glycol monoethyl ether
- Diethylene glycol mono-n-butyl ether
- Dipropyleneglycol methyl ether
- C12-15 pareth-12 a polyethylene glycol ether used as an emulsifier in cosmetics
- Dialkyl ethers
- Ethylene glycol dimethyl ether - a higher boiling alternative to diethyl ether and THF, also used as a solvent for polysaccharides, a reagent in organometallic chemistry and in some electrolytes of lithium batteries
- Diethylene glycol dimethyl ether
- Triethylene glycol dimethyl ether
- Tetraethylene glycol dimethyl ether
- Ethylene glycol diethyl ether
- Ethylene glycol dibutyl ether
- Esters
- Ethylene glycol methyl ether acetate
- Ethylene glycol monoethyl ether acetate
- Ethylene glycol monobutyl ether acetate
- Monoalkyl ethers
- Ethoxylates
- Laundry detergents and fabric care formulations[29]
- Dishwashing liquids and automatic dishwasher detergents[30]
- Household and institutional surface cleaners[31]
- Industrial degreasers and metal cleaning formulations[32]
- Emulsion polymerization aids and latex stabilization agents[33]
- Paints, coatings, and pigment dispersions[34]
- Textile wetting, scouring, and dyeing auxiliaries[35]
- Paper pulping, deinking, and paper coating formulations[36]
- Agricultural adjuvants in pesticide and herbicide formulations[37]
- Cosmetic and personal care emulsifiers and solubilizers[38]
- Pharmaceutical excipients and drug-delivery solubilizers[39]
- Food-contact processing aids and equipment cleaners[40]
- Oilfield chemicals and enhanced oil recovery surfactants[41]
- Polymer processing aids and plastic additive formulations[42]
- Leather processing and finishing agents[43]
- Emulsion polymer stabilizers for adhesives and sealants[44]
- Ethylene glycol – via ethylene oxide hydration
- 1,2-Dichloroethane
- Trichloroethylene
- Tetrachloroethylene – also called perchloroethylene; used as a dry cleaning solvent and degreaser
- Vinyl chloride – monomer for polyvinyl chloride
- Polyvinyl chloride (PVC) – a type of plastic
- Ethanol – via ethylene hydration (chemical reaction adding water)
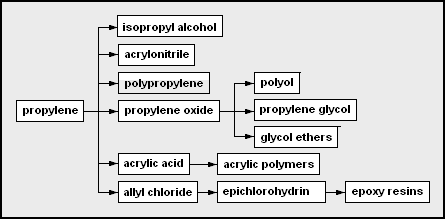
- Propylene – used as a monomer and a chemical feedstock
- Isopropyl alcohol – 2-propanol; often used as a solvent or rubbing alcohol
- Acrylonitrile – useful as a monomer in forming Orlon, ABS
- Polypropylene – polymerized propylene
- Propylene oxide[48]
- Polyether polyol – used in the production of polyurethanes
- Propylene glycol – used in engine coolant [49] and aircraft deicer fluid
- Glycol ethers – from the condensation of glycols
- Acrylic acid
- Allyl chloride
- Epichlorohydrin – chloro-oxirane; used in epoxy resin formation
- Epoxy resins – a type of polymerizing glue from bisphenol A, epichlorohydrin, and some amine
- Epichlorohydrin – chloro-oxirane; used in epoxy resin formation
- Butene
- Isomers of butylene – useful as monomers or co-monomers
- Isobutylene – feed for making methyl tert-butyl ether (MTBE) or monomer for copolymerization with a low percentage of isoprene to make butyl rubber
- 1,3-Butadiene (or buta-1,3-diene) – a diene often used as a monomer or co-monomer for polymerization to elastomers such as polybutadiene, styrene-butadiene rubber, or a plastic such as acrylonitrile-butadiene-styrene (ABS)
- Synthetic rubbers – synthetic elastomers made of any one or more of several petrochemical (usually) monomers such as 1,3-butadiene, styrene, isobutylene, isoprene, chloroprene; elastomeric polymers are often made with a high percentage of conjugated diene monomers such as 1,3-butadiene, isoprene, or chloroprene
- Isomers of butylene – useful as monomers or co-monomers
- Higher olefins
- Polyolefins – such poly-alpha-olefins, which are used as lubricants
- Alpha-olefins – used as monomers, co-monomers, and other chemical precursors. For example, a small amount of 1-hexene can be copolymerized with ethylene into a more flexible form of polyethylene.
- Other higher olefins
- Detergent alcohols
==Sand time==
- Han, Y. -F.; Wang, J. -H.; Kumar, D.; Yan, Z.; Goodman, D. W. (2005-06-10). "A kinetic study of vinyl acetate synthesis over Pd-based catalysts: kinetics of vinyl acetate synthesis over Pd–Au/SiO2 and Pd/SiO2 catalysts". Journal of Catalysis. 232 (2): 467–475. doi:10.1016/j.jcat.2005.04.001. ISSN 0021-9517.
- Flick, E. W. Adhesive and Sealant Compound Formulations. William Andrew Publishing, 1999.
- Harper, C. A. Handbook of Plastics, Elastomers, and Composites. McGraw-Hill, 2002.
- Encyclopedia Britannica. “Polyvinyl Acetate.”
- Odian, G. Principles of Polymerization. Wiley-Interscience, 2004.
- Harper, C. A. Handbook of Plastics, Elastomers, and Composites. McGraw-Hill, 2002.
- Odian, G. Principles of Polymerization. Wiley-Interscience, 2004.
- Flick, E. W. Adhesive and Sealant Compound Formulations. William Andrew Publishing, 1999.
- Harper, C. A. Handbook of Plastics, Elastomers, and Composites. McGraw-Hill, 2002.
- Encyclopedia Britannica. “Polyvinyl Acetate.”
- Horie, V. Materials for Conservation. Butterworth-Heinemann, 2010.
- Horie, V. Materials for Conservation. Butterworth-Heinemann, 2010.
- Odian, G. Principles of Polymerization. Wiley-Interscience, 2004.
- Harper, C. A. Handbook of Plastics, Elastomers, and Composites. McGraw-Hill, 2002.
- European Food Safety Authority. “Re-evaluation of Polyvinyl Acetate (E 1203) as a Food Additive.” EFSA Journal, 2010.
- Harper, C. A. Handbook of Plastics, Elastomers, and Composites. McGraw-Hill, 2002.
- Flick, E. W. Adhesive and Sealant Compound Formulations. William Andrew Publishing, 1999.
- . Holmberg, B. Jönsson, B. Kronberg, B. Lindman, ‘‘Surfactants and Polymers in Aqueous Solution’’. Wiley, 2002.
- D. Myers, ‘‘Surfactant Science and Technology’’, 3rd ed. Wiley, 2006.
- Tadros, ‘‘Applied Surfactants: Principles and Applications’’. Wiley-VCH, 2005.
- M. J. Rosen, J. T. Kunjappu, ‘‘Surfactants and Interfacial Phenomena’’, 4th ed. Wiley, 2012.
- D. Urban, K. Takamura, ‘‘Polymer Dispersions and Their Industrial Applications’’. Wiley-VCH, 2002.
- S. Paul, ‘‘Surface Coatings: Science and Technology’’. Wiley, 1985.
- W. D. Schindler, P. J. Hauser, ‘‘Chemical Finishing of Textiles’’. Woodhead Publishing, 2004.
- C. J. Biermann, ‘‘Handbook of Pulping and Papermaking’’. Academic Press, 1996.
- J. M. Green, G. B. Beestman, ‘‘Pesticide Formulations and Application Systems’’. ASTM International, 1990.
- A. O. Barel, M. Paye, H. I. Maibach, ‘‘Handbook of Cosmetic Science and Technology’’. CRC Press, 2014.
- A. T. Florence, D. Attwood, ‘‘Physicochemical Principles of Pharmacy’’. Pharmaceutical Press, 2016.
- FAO/WHO Joint Expert Committee on Food Additives, ‘‘Evaluation of Certain Food Additives: Surfactants and Processing Aids’’.
- L. L. Schramm, ‘‘Surfactants: Fundamentals and Applications in the Petroleum Industry’’. Cambridge University Press, 2000.
- G. Wypych, ‘‘Handbook of Plasticizers’’. ChemTec Publishing, 2017.
- A. D. Covington, ‘‘Tanning Chemistry’’. Royal Society of Chemistry, 2009.
- E. W. Flick, ‘‘Handbook of Adhesive Raw Materials’’. Noyes Publications, 1998.
- Lee, Eo Jin; Lee, Jong Won; Lee, Joongwon; Min, Hyung-Ki; Yi, Jongheop; Song, In Kyu; Kim, Do Heui (2018-06-01). "Ag-(Mo-W)/ZrO2 catalysts for the production of propylene oxide: Effect of pH in the preparation of ZrO2 support". Catalysis Communications. 111: 80–83. doi:10.1016/j.catcom.2018.04.005. ISSN 1566-7367. S2CID 103189174.
- HU patent 209546B, Forstner, Janos; Gal, Lajos & Feher, Pal et al., "Anti-freeze solution for internal combustion engines", published 1994-07-28