User:Karlhahn/oldSand
From Wikipedia, the free encyclopedia
Castner-Kellner process
The Castner-Kellner process is a method of electrolysis on an aqueous alkali chloride solution (usually sodium chloride solution) to produce the corresponding alkali hydroxide.
Process details

The aparatus shown is divided into two types of cells separated by slate walls. The first type, shown on the right and left of the diagram, uses an electrolyte of of sodium chloride solution, a graphite anode (A), and a mercury cathode (M). The other type of cell, shown in the center of the diagram, uses an electrolyte of sodium hydroxide solution, a mercury anode (M), and an iron cathode (D). Note that the mercury electrode is shared between the two cells. This is achieved by having the walls separating the cells dip below the level of the electrolytes but still allow the mercury to flow beneath them.
The reaction at anode (A) is:
- 2Cl– → Cl2 + 2e–
The chlorine gas that results vents at the top of the outside cells where it is collected as a byproduct of the process. The reaction at the mercury cathode in the outer cells is
- 2Na+ + 2e– → 2Na (amalgam)
The sodium metal formed by this reaction dissolves in the mercury to form an amalgam. The mercury conducts the current from the outside cells to the inside cell. In addition, a rocking mechanism (B shown by fulcrum on the left and rotating eccentric on the right) agitates the mercury to transport the dissolved sodium metal from the outside cells to the inside cell.
The anode reaction in the center cell takes place at the interface between the mercury and the sodium hydroxide solution.
- 2Na + 2e– → 2Na+
Finally at the iron cathode (D) of the center cell the reaction is
- 2H2O + 2e– → 2OH– + H2
The net effect is that the concentration of sodium chloride in the outside cells decreases and the concentration of sodium hydroxide in the center cell increases.
Extraction of Copper from its Ores
| Name | Formula | % Copper when pure |
|---|---|---|
| Chalcopyrite | CuFeS2 | 34.5 |
| Chalcocite | CuS | 79.8 |
| Bornite | 2Cu2S•CuS•FeS | 63.3 |
| Tetrahedrite | Cu3SbS3 + x(Fe,Zn)6Sb2S9 | 32-45 |
| Malachite | CuCO3•Cu(OH)2 | 57.3 |
| Azurite | 2CuCO3•Cu(OH)2 | 55.1 |
| Cuprite | Cu2O | 88.8 |
| Chrysocolla | CuO•SiO2•2H2O | 37.9 |
|
Principal Copper-bearing Minerals[1] | ||
Most copper ores contain one or more of the copper-bearing minerals shown in the table. Ores containing as little as 1% copper can be economically exploited. Ores are crushed then concentrated a combination of gravity separation and froth floatation. The resulting concentrate is about 30% copper, having extracted 90% of the copper in the original ore.
Most copper ores contain large amounts of sulfides. These ores are roasted prior to smelting. The smelting is done by reverberatory furnace, although blast furnaces were used in the past. At around 1500°C the charge separates into layers of slag (silicates) and matte (sulfides). This also has the effect of separating much of the iron content of the ore because copper preferentially collects in the matte layer while iron preferentially collects in the slag.
Copper mattes from the reverberatory furnace are then separated from the remaining iron and converted to metal by heating them in an air blow, which effects the following reactions:
- 2FeS + 3O2 → 2FeO + 2SO2
- 2Cu2S + 3O2 → 2Cu2O + 2SO2
- Cu2S + 2Cu2O → 6Cu + SO2
The addition of silica absorbs the FeO into a silicate slag layer. The remaining metal layer is poured off and cast as impure blister copper. Impurities are primarily arsenic, antimony, bismuth, and precious metals. To obtain copper pure enough for electrical products, the blister copper must be electrolytically refined. Anodes are cast from blister copper. Cathodes are made of thin rolled sheets of pure copper. The electrolyte is a 10-16% aqueous sulfuric acid solution with 3-4% dissoved copper(II) ions. The cell requires only a 0.2-0.4 volt potential. At the anode, copper and less noble metals dissolve. More noble metals such as silver and gold settle to the bottom of the cell as anode mud, which form a saleable byproduct. At the cathode, copper metal plates out but less noble metals remain in solution. The process results in copper that is 99.9% pure.[1]
Chlorine Compounds
| Oxidation state | Name | Forumula | Example compounds |
|---|---|---|---|
| –1 | chlorides | Cl– | ionic chlorides, organic chlorides, hydrochloric acid |
| 0 | chlorine | Cl2 | elemental chlorine |
| +1 | hypochlorites | ClO– | sodium and calcium hypochlorite |
| +3 | chlorites | ClO2– | sodium chlorite |
| +5 | chlorates | ClO3– | sodium chlorate, potassium chlorate |
| +7 | perchlorates | ClO4– | potassium perchlorate, perchloric acid, organic perchlorates, ammonium perchlorate, magnesium perchlorate |
Chlorine exists in all odd numbered oxidation states from –1 to +7, as well as the elemental state of zero. Progressing through the states, hydrogen chloride gas can be oxidized using manganese dioxide or catalytically by air to elemental chlorine gas. The solubility of chlorine in water is increased if the water contains dissolved alkali hydroxide. This is due to disproportionation:
- Cl2 + 2OH– → Cl– + ClO– + H2O
In hot concentrated alkali solution disproportionation continues:
- 2ClO– → Cl– + ClO2–
- ClO– + ClO2– → Cl– + ClO3–
Potassium chlorate can be crystalized from solutions formed by the above reactions. If its crystals are heated, it undergoes the final disproportionation step.
- ClO– + ClO3– → Cl– + ClO4–
This same progression from chloride to perchlorate can be accomplished by electrolysis. The anode reaction progression is:
- Cl– + H2O → ClO– + 2H+ + 2e–
- ClO– + H2O → ClO2– + 2H+ + 2e–
- ClO2– + H2O → ClO3– + 2H+ + 2e–
- ClO3– + H2O → ClO4– + 2H+ + 2e–
Each step is accompanied at the cathode by
- 2H+ + 2e– → H2
Ethanol Chemistry
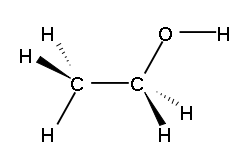
The chemistry of ethanol is largely that of its hydroxyl group.
- Acid-base chemistry
Ethanol's hydroxyl proton is weakly acidic, having a pKa of only 15.9, compared to water's 15.7[2] (Ka of ethanol is a measure of . Note that Ka of water is derived by dividing water's dissociation constant, moles²/liter, by its molar density of 55.5 moles/liter). Ethanol can be quantitatively converted to its conjugate base, the ethoxide ion (CH3CH2O−), by reaction with an alkali metal such as sodium. This reaction evolves hydrogen gas:
- Nucleophilic substitution
In aprotic solvents, ethanol reacts with hydrogen halides to produce ethyl halides such as ethyl chloride and ethyl bromide via nucleophilic substitution:
Ethyl halides can also be produced by reacting ethanol by more specialized halogenating agents, such as thionyl chloride for preparing ethyl chloride, or phosphorus tribromide for preparing ethyl bromide.
- Esterification
Under acid-catalysed conditions, ethanol reacts with carboxylic acids to produce ethyl esters and water:
- RCOOH + HOCH2CH3 → RCOOCH2CH3 + H2O
The reverse reaction, hydrolysis of the resulting ester back to ethanol and the carboxylic acid, limits the extent of reaction, and high yields are unusual unless water can be removed from the reaction mixture as it is formed. Esterification can also be carried out using more a reactive derivative of the carboxylic acid, such as an acyl chloride or acid anhydride. A very common ester of ethanol is ethyl acetate, found in nailpolish remover.
Ethanol can also form esters with inorganic acids. Diethyl sulfate and triethyl phosphate, prepared by reacting ethanol with sulfuric and phosphoric acid, respectively, are both useful ethylating agents in organic synthesis. Ethyl nitrite, prepared from the reaction of ethanol with sodium nitrite and sulfuric acid, was formerly a widely-used diuretic.
- Dehydration
Strong acids, such as sulfuric acid, can catalyse ethanol's dehydration to form either diethyl ether or ethylene:
- 2 CH3CH2OH → CH3CH2OCH2CH3 + H2O
Although sulfuric acid catalyses this reaction, the acid is diluted by the water that is formed, which makes the reaction inefficient. Which product, diethyl ether or ethylene, predominates depends on the precise reaction conditions.
- Oxidation
Ethanol can be oxidized to acetaldehyde, and further oxidized to acetic acid. In the human body, these oxidation reactions are catalysed by enzymes. In the laboratory, aqueous solutions of strong oxidizing agents, such as chromic acid or potassium permanganate, oxidize ethanol to acetic acid, and it is difficult to stop the reaction at acetaldehyde at high yield. Ethanol can be oxidized to acetaldehyde, without overoxidation to acetic acid, by reacting it with pyridinium chromic chloride.
- Combustion

Combustion of ethanol forms carbon dioxide and water:
- C2H5OH + 3 O2 → 2 CO2 + 3 H2O
Liquid/vapor equilibrium thermodynamic data
The table below gives thermodynamic data of liquid CO2 in equilibrium with its vapor at various temperatures. Heat content data, heat of vaporization, and entropy values are relative to the liquid state at 0°C temperature and 3483 kPa pressure. To convert heat values to molar values, multiply by 44.095 grams/mole. To convert densities to liters/mole, multiply by 22.678 liter-grams/cm3-mole. Data obtained from CRC Handbook of Chemistry and Physics, 44th ed. pages 2560-2561, except for critical temperature line (31.1°C) and temperatures –30°C and below, which are taken from Lange's Handbook of Chemistry, 10th ed. page 1463.
| Carbon dioxide liquid/vapor equilibrium thermodynamic data | ||||||||
| Temp. °C | Pvap Vapor pressure kPa | Hliq Heat content liquid J/gr | Hvap Heat content vapor J/gr | ΔvapH Heat of vapor- ization J/gr | ρvap Density of vapor gr/cm3 | ρliq Density of liquid gr/cm3 | Sliq Entropy liquid J/mol-°C | Svap Entropy vapor J/mol-°C |
|---|---|---|---|---|---|---|---|---|
| –56.6 | 518.3 | 1.179 | ||||||
| –56.0 | 531.8 | 1.177 | ||||||
| –54.0 | 579.1 | 1.169 | ||||||
| –52.0 | 629.6 | 1.162 | ||||||
| –50.0 | 683.4 | 1.155 | ||||||
| –48.0 | 740.6 | 1.147 | ||||||
| –46.0 | 801.3 | 1.139 | ||||||
| –44.0 | 865.6 | 1.131 | ||||||
| –42.0 | 933.8 | 1.124 | ||||||
| –40.0 | 1005.7 | 1.116 | ||||||
| –38.0 | 1081.6 | 1.108 | ||||||
| –36.0 | 1161.8 | 1.100 | ||||||
| –34.0 | 1246.2 | 1.092 | ||||||
| –32.0 | 1335.1 | 1.084 | ||||||
| -30.0 | 1428.6 | 1.075 | ||||||
| –28.89 | 1521 | –55.69 | 237.1 | 292.9 | 0.03846 | 1.0306 | –9.48 | 43.41 |
| –27.78 | 1575 | –53.76 | 237.3 | 291.0 | 0.03987 | 1.0276 | –9.13 | 43.21 |
| –26.67 | 1630 | –51.84 | 237.6 | 289.4 | 0.04133 | 1.0242 | –8.78 | 43.01 |
| –25.56 | 1686 | –49.87 | 237.6 | 287.5 | 0.04283 | 1.0209 | –8.45 | 42.78 |
| –24.44 | 1744 | –47.91 | 237.8 | 285.7 | 0.04440 | 1.0170 | –8.10 | 42.56 |
| –23.33 | 1804 | –45.94 | 237.8 | 283.6 | 0.04600 | 1.0132 | –7.75 | 42.36 |
| –22.22 | 1866 | –43.93 | 237.8 | 281.7 | 0.04767 | 1.0093 | –7.40 | 42.14 |
| –21.11 | 1928 | –41.92 | 237.8 | 279.6 | 0.04938 | 1.0053 | –7.05 | 42.94 |
| –20.00 | 1993 | –39.91 | 237.8 | 277.8 | 0.05116 | 1.0011 | –6.68 | 41.71 |
| –18.89 | 2059 | –37.86 | 237.8 | 275.7 | 0.05300 | 0.9968 | –6.31 | 41.49 |
| –17.78 | 2114 | –35.82 | 237.6 | 273.6 | 0.05489 | 0.9923 | –5.98 | 41.27 |
| –16.67 | 2197 | –33.73 | 237.6 | 271.2 | 0.05686 | 0.9875 | –5.61 | 41.05 |
| –15.56 | 2269 | –31.64 | 237.3 | 269.2 | 0.05888 | 0.9829 | –5.26 | 40.83 |
| –14.44 | 2343 | –29.54 | 237.3 | 266.9 | 0.06098 | 0.9782 | –4.91 | 40.61 |
| –13.33 | 2418 | –27.41 | 237.1 | 264.5 | 0.06314 | 0.9734 | –4.54 | 40.39 |
| –12.22 | 2495 | –25.27 | 236.9 | 262.2 | 0.06539 | 0.9665 | –4.17 | 40.15 |
| –11.11 | 2574 | –23.09 | 236.7 | 259.7 | 0.06771 | 0.9639 | –3.80 | 39.92 |
| –10.00 | 2654 | –20.90 | 236.4 | 257.3 | 0.07011 | 0.9592 | –3.43 | 39.68 |
| –8.89 | 2738 | –18.69 | 235.9 | 254.8 | 0.07259 | 0.9543 | –3.06 | 39.46 |
| –7.78 | 2823 | –16.45 | 235.7 | 252.2 | 0.07516 | 0.9494 | –2.69 | 39.22 |
| –6.67 | 2910 | –14.18 | 235.2 | 249.4 | 0.07783 | 0.9443 | –2.32 | 38.98 |
| –5.56 | 2999 | –11.90 | 234.8 | 246.6 | 0.08059 | 0.9393 | –1.94 | 38.74 |
| –4.44 | 3090 | –9.977 | 234.3 | 243.8 | 0.08347 | 0.9340 | –1.57 | 38.50 |
| –3.89 | 3136 | –8.410 | 234.1 | 242.4 | 0.08494 | 0.9313 | –1.37 | 38.37 |
| –2.78 | 3230 | –6.046 | 233.6 | 239.7 | 0.08797 | 0.9260 | –0.98 | 38.12 |
| –1.67 | 3327 | –3.648 | 232.9 | 236.6 | 0.09111 | 0.9206 | –0.59 | 37.88 |
| –0.56 | 3425 | –1.222 | 232.4 | 233.6 | 0.09438 | 0.9150 | –0.20 | 37.62 |
| 0.56 | 3526 | 1.234 | 231.7 | 230.5 | 0.09776 | 0.9094 | 0.20 | 37.36 |
| 1.67 | 3629 | 3.728 | 231.0 | 227.3 | 0.1013 | 0.9036 | 0.61 | 37.08 |
| 2.78 | 3735 | 6.268 | 230.4 | 224.0 | 0.1050 | 0.8975 | 1.01 | 36.83 |
| 3.89 | 3981 | 8.445 | 229.4 | 220.5 | 0.1088 | 0.8914 | 1.42 | 36.55 |
| 5.00 | 3953 | 11.46 | 228.5 | 217.0 | 0.1128 | 0.8850 | 1.83 | 36.25 |
| 6.11 | 4067 | 14.13 | 227.6 | 213.4 | 0.1169 | 0.8784 | 2.25 | 35.98 |
| 7.22 | 4182 | 16.85 | 226.5 | 209.7 | 0.1213 | 0.8716 | 2.69 | 35.68 |
| 8.33 | 4300 | 19.63 | 225.4 | 205.8 | 0.1258 | 0.8645 | 3.12 | 35.39 |
| 9.44 | 4420 | 22.46 | 224.3 | 201.8 | 0.1306 | 0.8571 | 3.56 | 35.07 |
| 10.56 | 4544 | 25.36 | 223.1 | 197.7 | 0.1355 | 0.8496 | 4.02 | 34.76 |
| 11.67 | 4670 | 28.33 | 221.8 | 193.4 | 0.1408 | 0.8418 | 4.48 | 34.45 |
| 12.78 | 4798 | 31.35 | 220.3 | 188.9 | 0.1463 | 0.8338 | 4.94 | 34.11 |
| 13.89 | 4929 | 34.49 | 218.8 | 184.3 | 0.1521 | 0.8254 | 5.42 | 33.76 |
| 15.00 | 5063 | 37.30 | 217.2 | 179.5 | 0.1583 | 0.8168 | 5.92 | 33.41 |
| 16.11 | 5200 | 41.03 | 215.1 | 174.4 | 0.1648 | 0.8076 | 6.42 | 33.02 |
| 17.22 | 5340 | 44.48 | 213.6 | 169.1 | 0.1717 | 0.7977 | 6.96 | 32.66 |
| 18.33 | 5482 | 48.03 | 211.5 | 163.5 | 0.1791 | 0.7871 | 7.49 | 32.25 |
| 19.44 | 5628 | 51.71 | 209.4 | 157.6 | 0.1869 | 0.7759 | 8.04 | 31.83 |
| 20.56 | 5776 | 55.61 | 207.0 | 151.4 | 0.1956 | 0.7639 | 8.63 | 31.38 |
| 21.67 | 5928 | 59.66 | 204.3 | 144.7 | 0.2054 | 0.7508 | 9.24 | 30.90 |
| 22.78 | 6083 | 63.97 | 201.5 | 137.5 | 0.2151 | 0.7367 | 9.89 | 30.39 |
| 23.89 | 6240 | 68.58 | 198.4 | 129.8 | 0.2263 | 0.7216 | 10.57 | 29.85 |
| 25.00 | 6401 | 73.51 | 194.8 | 121.3 | 0.2387 | 0.7058 | 11.31 | 29.24 |
| 26.11 | 6565 | 78.91 | 190.7 | 111.8 | 0.2532 | 0.6894 | 12.10 | 28.60 |
| 27.22 | 6733 | 84.94 | 186.0 | 101.1 | 0.2707 | 0.6720 | 12.99 | 27.84 |
| 28.33 | 6902 | 91.88 | 180.4 | 88.49 | 0.2923 | 0.6507 | 14.00 | 26.95 |
| 29.44 | 7081 | 100.4 | 173.1 | 72.72 | 0.3204 | 0.6209 | 15.24 | 25.85 |
| 30.00 | 7164 | 105.6 | 168.4 | 62.76 | 0.3378 | 0.5992 | 16.01 | 25.15 |
| 30.56 | 7253 | 112.3 | 162.3 | 50.04 | 0.3581 | 0.5661 | 16.99 | 24.24 |
| 31.1 | 7391 | 0.00 | 0.4641 | 0.4641 | ||||
| Temp. °C | Pvap Vapor pressure kPa | Hliq Heat content liquid J/gr | Hvap Heat content vapor J/gr | ΔvapH Heat of vapor- ization J/gr | ρvap Density of vapor gr/cm3 | ρliq Density of liquid gr/cm3 | Sliq Entropy liquid J/mol-°C | Svap Entropy vapor J/mol-°C |
from [Surface tension]]
Water Strider Physics
The photograph shows water striders standing on the surface of a pond. It is clearly visible that their feet cause indentations in the water's surface. It is intuitively evident that the surface with indentations has more surface area than a flat surface. If surface tension tends to minimize surface area, how is it that the water striders are increasing the surface area?
Recall that what nature really tries to minimize is potential energy. By increasing the surface area of the water, the water striders have increased the potential energy of that surface. But note also that the water striders' center of mass is lower than it would be if they were standing on a flat surface. So their potential energy is decreased. Indeed when you combine the two effects, the net potential energy is minimized. If the water striders depressed the surface any more, the increased surface energy would more than cancel the reduction in the decreased energy of lowering the insects' center of mass. If they depressed the surface any less, their higher center of mass would more than cancel the reduction in surface energy.
The photo of the water striders also illustrates the notion of surface tension being like having an elastic film over the surface of the liquid. In the surface depressions at their feet it is easy to see that the reaction of that imagined elastic film is exactly countering the weight of the insects.
Liquid in a Cylindrical Tube
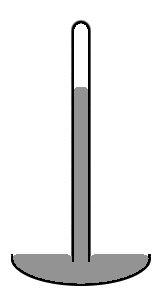
An old style mercury barometer consists of a vertical glass tube about 1 cm in diameter partially filled with mercury, and with a vacuum in the unfilled volume (see diagram to the left). Notice that the mercury level at the center of the tube is higher than at the at the edges, making the upper surface of the mercury dome-shaped. The center of mass of the entire column of mercury would be slightly lower if the top surface of the mercury were flat over the entire crossection of the tube. But the dome-shaped top gives slightly less surface area to the entire mass of mercury. Again the two effects combine to minimize the total potential energy. Such a surface shape is known as a convex meniscus.
The reason we consider the surface area of the entire mass of mercury, including the part of the surface that is in contact with the glass is because mercury does not adhere at all to glass. So the surface tension of the mercury acts over its entire surface area, including where it is in contact with the glass. If instead of glass, the tube were made out of copper, the situation would be very different. Mercury aggressively adheres to copper. So in a copper tube, the level of mercury at the center of the tube will be lower rather than higher than at the edges (that is, it would be a concave meniscus). In a situation where the liquid adheres to the walls of its container, we consider the part of the fluid's surface area that is in contact with the container to have negative surface tension. The fluid then works to maximize the contact surface area. So in this case increasing the area in contact with the container decreases rather than increases the potential energy. That decrease is enough compensate for the increase potential energy associated with the lifting of the fluid near the walls of the container.
Pool of Liquid on a Nonadhesive Surface
Pouring mercury onto a horizontal flat sheet of glass results in a puddle that has a perceptible thickness (do not try this except under a fume hood. Mercury vapor is a toxic hazard). The puddle will only spread out only to the point where it is a little under a centimeter thick, and no thinner. Again this is the action of mercury's strong surface tension. The liquid mass flattens out because that brings as much of the mercury to as low a level as possible. But the surface tension, at the same time, is acting to reduce the total surface area. The result is the compromise of a puddle of a nearly fixed thickness.
The same surface tension demonstration can be done with water, but only on a surface made of a substance that the water does not adhere to. Candle wax is such a substance. Water poured onto a smooth, flat, horizontal wax surface will behave similarly to the mercury poured onto glass.
Descriptive chemistry (lead)
Various oxidized forms of lead are easily reduced to the metal. An example is heating PbO with mild organic reducing agents such as glucose. A mixuture of the oxide and the sulfide heated together without any reducing agent will also form the metal.[3]
- 2PbO + PbS → 3 Pb + SO2
Metallic lead is attacked only superficially by air, forming a thin layer of oxide that protects it from further oxidation. The metal is not attacked by sulfuric or hydrochloric acids. It does, however, dissovle in nitric acid with the evolution of nitric oxide gas to form dissolved Pb(NO3)2.
- 3 Pb + 8 H+ + 8 NO3– → 3 Pb2+ + 6 NO3– + 2 NO + 4H2O
When heated with nitrates of alkali metals, metallic lead oxidizes to form PbO (also known as litharge), leaving the corresponding alkali nitrite. PbO is representative of lead's II oxidation state. It is soluble in nitric and acetic acids, from which solutions it is possible to preciptate halide, sulfate, and basic carbonate salts of lead. The sulfide can also be precipitated from acetate solutions. These salts are all poorly soluble in water. Among the halides, the iodide is less soluble than the bromide, which, in turn, is less soluble than the chloride.[4]
The II oxide is also soluble in alkali hydroxide solutions to form the corresponding plumbite salt.[3]
- PbO + 2OH– + H2O → Pb(OH)42–
Chlorination of plumbite solutions causes the formation of lead's IV oxidation state. Lead dioxide is representative of the IV state, and is a powerful oxidizing agent. The chloride of this oxidation state is formed only with difficulty and decomposes readily into the II chloride and chlorine gas. The bromide and iodide of IV lead are not known to exist.[4] Lead dioxide dissolves in alkali hydroxide solutions to form the corresponding plumbates.[3]
- PbO2 + 2 OH– + 2 H2O → Pb(OH)62–
Rewrite Project
for calcium carbonate water solubility section:
Original version
Calcium carbonate is not rigorously insoluble in water. For the following equilibrium reaction
- CaC03(solid) ↔ Ca2+ + CO32−, we take a solubility product at 25°C (Ksp=3.8 x 10−9 is given in [5]
Considering a saturated pure CaCO3 solution, the calculation of the Ca2+ concentration must take into account the equilibria between the three different carbonate forms (H2CO3, HCO3− and CO32−) as well as the equilibrium between H2CO3 and the dissolved CO2 and the equilibrium between the dissolved CO2 and the gaseous CO2 above the solution. The reactions involved are the following (see carbonic acid):
- CO2(gas) ↔ CO2(dissolved) with where k'c=29.76 atm/(mol/L) at 25°C (Henry constant), being the CO2 partial pressure.
- CO2(dissolved) + H2O ↔ H2CO3 with at 25°C
- H2CO3 ↔ H+ + HCO3− with at 25°C
- HCO3− ↔ H+ + CO32− with at 25°C
The above relations (together with the relation and the neutrality condition , i.e. 7 equations for 7 unknowns) allow the numerical calculation of the pH and of the Ca2+ concentration as a function of . The result is given in the table below:
| (atm) | pH | [Ca2+] (mol/L) |
| 10−12 | 12.0 | 5.19 x 10−3 |
| 10−10 | 11.3 | 1.12 x 10−3 |
| 10−8 | 10.7 | 2.55 x 10−4 |
| 10−6 | 9.83 | 1.20 x 10−4 |
| 10−4 | 8.62 | 3.16 x 10−4 |
| 3.5 x 10−4 | 8.27 | 4.70 x 10−4 |
| 10−3 | 7.96 | 6.62 x 10−4 |
| 10−2 | 7.30 | 1.42 x 10−3 |
| 10−1 | 6.63 | 3.05 x 10−3 |
| 1 | 5.96 | 6.58 x 10−3 |
| 10 | 5.30 | 1.42 x 10−2 |
We see that for normal atmospheric conditions ( atm), we get a slightly basic solution (pH = 8.3) with a low Ca2+ concentration (4.7 x 10−4 mol/L i.e. 0.019 g/L of Ca). Increasing the CO2 pressure makes the solution slightly acid with a better Ca solubility (0.57 g/L of Ca at 10 atm). For decreasing CO2 pressure values, the solubility goes to a minimum for atm and then increases again as the solution gets strongly basic.
Remark: For > 10−4 atm, CO32−, H+ and OH− concentrations can be neglected in the neutrality condition. This means physically that we have essentially a calcium bicarbonate solution. In this case, the system can be solved analytically, giving (with a very good precision)
Current state of rewritten version
Calcium carbonate is poorly soluble in water. The equilibrium of its solution is given by the equation (with dissolved calcium carbonate on the right):
CaCO3 ⇋ Ca2+ + CO32– Ksp = 3.7×10–9 to 8.7×10–9 at 25 °C
where the solubility product for [Ca2+][CO32–] is given as anywhere from Ksp = 3.7×10–9 to Ksp = 8.7×10–9 at 25 °C, depending upon the data source.[6][7] What the equation means is that the product of number of moles of dissolved Ca2+ with the number of moles of dissolved CO32– cannot exceed the value of Ksp. This seemingly simple solubility equation, however, must be taken along with the more complicated equilibrium of carbon dioxide with water. Some of the CO32– combines with H+ in the solution according to:
HCO3– ⇋ H+ + CO32– Ka2 = 5.61×10–11 at 25 °C
HCO3– is known as the bicarbonate ion. Calcium bicarbonate is many times more soluble in water than calcium carbonate -- indeed it exists only in solution.
Some of the HCO3– combines with H+ in solution according to:
H2CO3 ⇋ H+ + HCO3– Ka1 = 2.5×10–4 at 25 °C
Some of the H2CO3 breaks up into water and dissolved carbon dioxide according to:
H2O + CO2(dissolved) ⇋ H2CO3 Kh = 1.70×10–3 at 25 °C
And dissolved carbon dioxide is in equilibrium with atmospheric carbon dioxide according to:
where k'c = 29.76 atm/(mol/L) at 25°C (Henry constant), being the CO2 partial pressure.
| Calcium Ion Solubility as a function of CO2 partial pressure | ||
|---|---|---|
| (atm) | pH | [Ca2+] (mol/L) |
| 10−12 | 12.0 | 5.19 × 10−3 |
| 10−10 | 11.3 | 1.12 × 10−3 |
| 10−8 | 10.7 | 2.55 × 10−4 |
| 10−6 | 9.83 | 1.20 × 10−4 |
| 10−4 | 8.62 | 3.16 × 10−4 |
| 3.5 × 10−4 | 8.27 | 4.70 × 10−4 |
| 10−3 | 7.96 | 6.62 × 10−4 |
| 10−2 | 7.30 | 1.42 × 10−3 |
| 10−1 | 6.63 | 3.05 × 10−3 |
| 1 | 5.96 | 6.58 × 10−3 |
| 10 | 5.30 | 1.42 × 10−2 |
For ambient air, is around 3.5×10–4 atmospheres (or equivalently 35 Pa). The last equation above fixes the concentration of dissolved CO2 as a function of , independent of the concentration of dissolved CaCO3. At atmospheric partial pressure of CO2, dissolved CO2 concentration is 1.2×10–5 moles/liter. The equation before that fixes the concentration of H2CO3 as a function of [CO2]. For [CO2]=1.2×10–5, it results in [H2CO3]=2.0×10–8 moles per liter. When [H2CO3] is known, the remaining three equations together with
H2O ⇋ H+ + OH– K = 10–14 at 25 °C
(which is true for all aqueous solutions), and the fact that the solution must be electrically neutral,
- 2[Ca2+] + [H+] = [HCO3–] + 2[CO32–] + [OH–]
make it possible to solve simultaneously for the remaining five unknown concentrations. The table on the right shows the solution for [Ca2+] and [H+] (in the form of pH) as a function of ambient partial pressure of CO2. At atmopheric levels of ambient CO2 the table indicates the solution will be slightly alkaline. The trends the table shows are
- 1) As ambient CO2 partial pressure is reduced below atmospheric levels, the solution becomes more and more alkaline. At extremly low , dissolved CO2, bicarbonate ion, and carbonate ion largely evaporate from the solution, leaving a highly alkaline solution of calcium hydroxide, which is more soluble than CaCO3.
- 2) As ambient CO2 partial pressure increases to levels above atmospheric, pH drops, and much of the carbonate ion is converted to bicarbonate ion, which results in higher solubility of Ca2+.
The effect of the latter is especially evident in day to day life of people who have hard water. Water in aquifers underground can be exposed to levels of CO2 much higher than atmospheric. As such water perculates through calcium carbonate rock, the CaCO3 dissolves according to the second trend. When that same water then water emerges from the tap, in time it comes into equilibrium with CO2 levels in the air by outgassing its excess CO2. The calcium carbonate becomes less soluble as a result and the excess precipitates as lime scale. This same process is responsible for the formation of stalactites and stalagmites in limestone caves.
Liquid/Vapor Equilibrium Data
| Vapor over Anhydrous Ammonia | ||||
|---|---|---|---|---|
| Temp. | Pressure | ρ of liquid | ρ of vapor | ΔvapH |
| –78 °C | 5.90 kPa | |||
| –75 °C | 7.93 kPa | 0.73094 g/cm3 | 7.8241×10–5 g/cm3 | |
| –70 °C | 10.92 kPa | 0.72527 g/cm3 | 1.1141×10–4 g/cm3 | |
| –65 °C | 15.61 kPa | 0.71953 g/cm3 | 1.5552×10–4 g/cm3 | |
| –60 °C | 21.90 kPa | 0.71378 g/cm3 | 2.1321×10–4 g/cm3 | |
| –55 °C | 30.16 kPa | 0.70791 g/cm3 | 2.8596×10–4 g/cm3 | |
| –50 °C | 40.87 kPa | 0.70200 g/cm3 | 3.8158×10–4 g/cm3 | 1417 J/g |
| –45 °C | 54.54 kPa | 0.69604 g/cm3 | 4.9940×10–4 g/cm3 | 1404 J/g |
| –40 °C | 71.77 kPa | 0.68999 g/cm3 | 6.4508×10–4 g/cm3 | 1390 J/g |
| –35 °C | 93.19 kPa | 0.68385 g/cm3 | 8.2318×10–4 g/cm3 | 1375 J/g |
| –30 °C | 119.6 kPa | 0.67764 g/cm3 | 1.0386×10–3 g/cm3 | 1361 J/g |
| –25 °C | 151.6 kPa | 0.67137 g/cm3 | 1.2969×10–3 g/cm3 | 1345 J/g |
| –20 °C | 190.2 kPa | 0.66503 g/cm3 | 1.6039×10–3 g/cm3 | 1330 J/g |
| –15 °C | 236.3 kPa | 0.65854 g/cm3 | 1.9659×10–3 g/cm3 | 1314 J/g |
| –10 °C | 290.8 kPa | 0.65198 g/cm3 | 2.3874×10–3 g/cm3 | 1297 J/g |
| –5 °C | 354.8 kPa | 0.64533 g/cm3 | 2.8827×10–3 g/cm3 | 1280 J/g |
| 0 °C | 429.4 kPa | 0.63857 g/cm3 | 3.4528×10–3 g/cm3 | 1263 J/g |
| 5 °C | 515.7 kPa | 0.63167 g/cm3 | 4.1086×10–3 g/cm3 | 1245 J/g |
| 10 °C | 614.9 kPa | 0.62469 g/cm3 | 4.8593×10–3 g/cm3 | 1226 J/g |
| 15 °C | 728.3 kPa | 0.61755 g/cm3 | 5.7153×10–3 g/cm3 | 1207 J/g |
| 20 °C | 857.1 kPa | 0.61028 g/cm3 | 6.6876×10–3 g/cm3 | 1187 J/g |
| 25 °C | 1003 kPa | 0.60285 g/cm3 | 7.7882×10–3 g/cm3 | 1167 J/g |
| 30 °C | 1166 kPa | 0.59524 g/cm3 | 9.0310×10–3 g/cm3 | 1146 J/g |
| 35 °C | 1350 kPa | 0.58816 g/cm3 | 1.0431×10–2 g/cm3 | 1124 J/g |
| 40 °C | 1554 kPa | 0.57948 g/cm3 | 1.2006×10–2 g/cm3 | 1101 J/g |
| 45 °C | 1781 kPa | 0.57130 g/cm3 | 1.3775×10–2 g/cm3 | 1083 J/g |
| 50 °C | 2032 kPa | 0.56287 g/cm3 | 1.5761×10–2 g/cm3 | 1052 J/g |
| 55 °C | 2310 kPa | 0.55420 g/cm3 | ||
| 60 °C | 2613 kPa | 0.54523 g/cm3 | 2.05×10–2 g/cm3 | |
| 65 °C | 2947 kPa | 0.53596 g/cm3 | ||
| 70 °C | 3312 kPa | 0.52632 g/cm3 | 2.65×10–2 g/cm3 | |
| 75 °C | 3711 kPa | 0.51626 g/cm3 | ||
| 80 °C | 4144 kPa | 0.50571 g/cm3 | 3.41×10–2 g/cm3 | |
| 85 °C | 4614 kPa | 0.49463 g/cm3 | ||
| 90 °C | 5123 kPa | 0.48290 g/cm3 | 4.39×10–2 g/cm3 | |
| 95 °C | 5672 kPa | 0.47041 g/cm3 | ||
| 100 °C | 6264 kPa | 0.45693 g/cm3 | 5.68×10–2 g/cm3 | |
The table above gives properties of the vapor/liquid equilibrium of anhydrous ammonia at various temperatures. The second column is vapor pressure in kPa. The third column is the density of the liquid phase. The fourth column is the density of the vapor. The fifth column is the heat of vaporization needed to convert one gram of liquid to vapor.
Equilibrium of vapor over aqueous solution
| Vapor over Aqueous Ammonia Solution[8] | |||
|---|---|---|---|
| Temp. | %wt NH3 | Partial Pressure NH3 |
Partial Pressure H2O |
| 0 °C | 4.72 | 1.52 kPa | 0.68 kPa |
| 9.15 | 3.31 kPa | 0.71 kPa | |
| 14.73 | 6.84 kPa | 0.55 kPa | |
| 19.62 | 11.0 kPa | 0.40 kPa | |
| 22.90 | 14.9 kPa | 0.37 kPa | |
| 10 °C | 4.16 | 2.20 kPa | 1.21 kPa |
| 8.26 | 4.96 kPa | 1.17 kPa | |
| 12.32 | 8.56 kPa | 1.01 kPa | |
| 15.88 | 12.68 kPa | 0.93 kPa | |
| 20.54 | 19.89 kPa | 0.83 kPa | |
| 21.83 | 22.64 kPa | 0.73 kPa | |
| 19.9 °C | 4.18 | 3.65 kPa | 2.19 kPa |
| 6.50 | 6.11 kPa | 2.15 kPa | |
| 6.55 | 6.13 kPa | 2.13 kPa | |
| 7.72 | 7.49 kPa | 2.08 kPa | |
| 10.15 | 10.75 kPa | 2.01 kPa | |
| 10.75 | 11.51 kPa | 1.96 kPa | |
| 16.64 | 22.14 kPa | 1.72 kPa | |
| 19.40 | 28.74 kPa | 1.64 kPa | |
| 23.37 | 40.32 kPa | 1.37 kPa | |
| 30.09 °C | 3.93 | 5.49 kPa | 4.15 kPa |
| 7.43 | 11.51 kPa | 3.89 kPa | |
| 9.75 | 16.00 kPa | 3.80 kPa | |
| 12.77 | 23.33 kPa | 3.55 kPa | |
| 17.76 | 38.69 kPa | 3.31 kPa | |
| 17.84 | 38.81 kPa | 3.24 kPa | |
| 21.47 | 53.94 kPa | 2.95 kPa | |
| 40 °C | 3.79 | 8.15 kPa | 7.13 kPa |
| 7.36 | 17.73 kPa | 6.76 kPa | |
| 11.06 | 29.13 kPa | 6.55 kPa | |
| 15.55 | 47.14 kPa | 5.52 kPa | |
| 17.33 | 57.02 kPa | ||
| 20.85 | 76.81 kPa | 5.04 kPa | |
| 50 °C | 3.29 | 10.54 kPa | 11.95 kPa |
| 5.90 | 20.17 kPa | 11.61 kPa | |
| 8.91 | 32.88 kPa | 11.07 kPa | |
| 11.57 | 45.56 kPa | 10.75 kPa | |
| 14.15 | 60.18 kPa | 10.27 kPa | |
| 14.94 | 64.94 kPa | 10.03 kPa | |
| 60 °C | 3.86 | 18.25 kPa | 19.21 kPa |
| 5.77 | 28.78 kPa | ||
| 7.78 | 40.05 kPa | 18.47 kPa | |
| 9.37 | 50.09 kPa | 18.07 kPa | |
| 9.37 | 63.43 kPa | 17.39 kPa | |
Hebrew Calendar
Summary of Calendar Calculations
The audience for this section is computer programmers who wish to write software that accurately computes dates in the Hebrew calendar. The following details are sufficient to generate such software.
- 1) The Hebrew calendar is computed by lunations. One lunation is reckoned at 29 days, 12 hours, 44 minutes, 3⅓ seconds, or equivalently 765433 halakim = 29 days, 13753 halakim.
- 2) A common year must be either 353, 354, or 355 days; a leap year must be 383, 384, or 385 days. A 353 or 383 day year is called kesidrah. A 354 or 384 day year is shelemah. A 355 or 385 day year is haserah.
- 3) Leap years follow a 19 year schedule in which years 3, 6, 8, 11, 14, 17, and 19 are leap years. The Jewish year 5752 (which starts in Gregorian year 1991) is the first year of a cycle.
- 4) 19 years is the same as 235 lunations.
- 5) The months are Tishri, Heshvan, Kislev, Tebeth, Shebhat, Adar, Nisan, Iyyar, Sivan, Tammuz, Av, and Elul. In addition, a second Adar (also called Veadar, Adar II, or Adar Sheni) is added in leap years. When added, it follows Adar.
- 6) Each month has either 29 or 30 days. A 30 day month is male, a 29 day month is haser.
- Nisan, Sivan, Av, Tishri, and Shebhat are always male.
- Iyyar, Tammuz, Elul, Tebeth, and Adar II are always haser.
- Adar is male in leap years, haser in common years.
- Heshvan and Kislev vary, but when they differ, Heshvan is haser and Kislev is male.
- 7) Tishri 1st (Rosh Hashana) is the day of a molad (new moon) unless certain conditions (dahiyyah sing; dahiyyot pl) exist.
- a) This dahiyyah exists whenever Tishri 10 (Yom Kippur) would fall on a Friday or a Sunday, or if Tishri 21 (7th day of Sukkot) would fall on a Saturday. This is equivalent to the molad being on Sunday, Wednesday, or Friday. Whenever this happens, Tishri 1 is delayed by 1 day.
- b) This dahiyyah exists whenever the molad occurs on or after noon. When this dahiyyah exists, Tishri 1 is delayed by 1 day. If this causes dahiyyah A to exist, Tishri 1 is delayed an additional day.
- c) If the year is to be a common year and the molad falls on a Tuesday on or after 3:11:20 am (3 hours 204 halakim) Jerusalem time, Tishri 1 is delayed by 2 days. This is because if it weren't delayed, the resulting year would be 356 days long.
- d) If the new year follows a leap year and the molad is on a Monday on or after 9:32:43 and one third seconds (9 hours 589 halakim), Tishri 1 is delayed 1 day. This is because if it weren't, the preceding year would have only 382 days.
- 8) Delays are implemented by adding a day to Kislev of the preceding year, making it male. If Kislev is already male, the day is added to Heshvan of the preceding year, making it male also. If a delay of 2 days is called for, both Heshvan and Kislev of the preceding year become male.
- 9) The molad of 08-Sep-1991, which is Rosh Hashana of Hebrew yer, 5752, is Julian day, 2448509 plus 3294 halakim.
Future Project: Tide Prediction
Plan to explain the method of harmonic constituents as detailed in U.S. Govt. Special Publication 92.
Reorganize Heat Capacity Ratio Table
| Heat Capacity Ratio for various gases[9][10] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Temp. | Gas | γ | Temp. | Gas | γ | Temp. | Gas | γ | ||
| –181°C | H2 | 1.597 | –76°C | H2 | 1.453 | 20°C | H2 | 1.41 | ||
| 100°C | H2 | 1.404 | 400°C | H2 | 1.387 | 1000°C | H2 | 1.358 | ||
| 2000°C | H2 | 1.318 | 20°C | He | 1.66 | 20°C | H2O | 1.33 | ||
| 100°C | H2O | 1.324 | 200°C | H2O | 1.310 | –180°C | Ar | 1.76 | ||
| 20°C | Ar | 1.67 | 0°C | Dry Air | 1.403 | 20°C | Dry Air | 1.40 | ||
| 100°C | Dry Air | 1.401 | 200°C | Dry Air | 1.398 | 400°C | Dry Air | 1.393 | ||
| 1000°C | Dry Air | 1.365 | 1400°C | Dry Air | 1.341 | 2000°C | Dry Air | 1.088 | ||
| 0°C | CO2 | 1.310 | 20°C | CO2 | 1.30 | 100°C | CO2 | 1.281 | ||
| 400°C | CO2 | 1.235 | 1000°C | CO2 | 1.195 | 20°C | CO | 1.40 | ||
| –181°C | O2 | 1.45 | –76°C | O2 | 1.415 | 20°C | O2 | 1.40 | ||
| 100°C | O2 | 1.399 | 200°C | O2 | 1.397 | 400°C | O2 | 1.394 | ||
| 20°C | NO | 1.40 | 20°C | N2O | 1.31 | –181°C | N2 | 1.47 | ||
| 15°C | N2 | 1.404 | 20°C | Cl2 | 1.34 | –115°C | CH4 | 1.41 | ||
| –74°C | CH4 | 1.35 | 20°C | CH4 | 1.32 | 19°C | Ne | 1.64 | ||
| 19°C | Kr | 1.68 | 19°C | Xe | 1.66 | 15°C | SO2 | 1.29 | ||
| 360°C | Hg | 1.67 | ||||||||
| Heat Capacity Ratio for various gases[11][12] | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Temp. | Gas | γ | Temp. | Gas | γ | Temp. | Gas | γ | ||
| –181°C | H2 | 1.597 | 200°C | Dry Air | 1.398 | 20°C | NO | 1.40 | ||
| –76°C | 1.453 | 400°C | 1.393 | 20°C | N2O | 1.31 | ||||
| 20°C | 1.41 | 1000°C | 1.365 | –181°C | N2 | 1.47 | ||||
| 100°C | 1.404 | 2000°C | 1.088 | 15°C | 1.404 | |||||
| 400°C | 1.387 | 0°C | CO2 | 1.310 | 20°C | Cl2 | 1.34 | |||
| 1000°C | 1.358 | 20°C | 1.30 | –115°C | CH4 | 1.41 | ||||
| 2000°C | 1.318 | 100°C | 1.281 | –74°C | 1.35 | |||||
| 20°C | He | 1.66 | 400°C | 1.235 | 20°C | 1.32 | ||||
| 20°C | H2O | 1.33 | 1000°C | 1.195 | 15°C | NH3 | 1.310 | |||
| 100°C | 1.324 | 20°C | CO | 1.40 | 19°C | Ne | 1.64 | |||
| 200°C | 1.310 | –181°C | O2 | 1.45 | 19°C | Xe | 1.66 | |||
| –180°C | Ar | 1.76 | –76°C | 1.415 | 19°C | Kr | 1.68 | |||
| 20°C | 1.67 | 20°C | 1.40 | 15°C | SO2 | 1.29 | ||||
| 0°C | Dry Air | 1.403 | 100°C | 1.399 | 360°C | Hg | 1.67 | |||
| 20°C | 1.40 | 200°C | 1.397 | 15°C | C2H6 | 1.22 | ||||
| 100°C | 1.401 | 400°C | 1.394 | 16°C | C3H8 | 1.13 | ||||
Translating Water (data page) equivalent from German
Current version of translation
Physical and Thermodynamic Tables
In the following tables, values are temperature dependent and to a lesser degree pressure dependent, and are arranged by state of aggregation (s=solid, lq=liquid, g=gas), which are clearly a function of temperature and pressure. All of the data were computed from data given in "Formulation of the Thermodynamic Properties of Ordinary Water Substance for Scientific and General Use" (1984). This applies to:
- T - temperature in degrees celsius
- V - specific volume in decimeter3 per kilogram
- H - specific enthalpy in kJ per kilogram
- U - internal energy in kJ per kilogram
- S - specific entropy in kJ per kilogram-Kelvin
- cp - specific (constant pressure) heat capacity in kJ per kilogram-Kelvin
- γ - Thermal expansion coefficient as 10–3 per Kelvin
- λ - Heat conductivity in milliwatt per meter-Kelvin
- η - Viscosity in μPa-seconds
- σ - Surface Tension millinewtons per meter
Standard conditions
In the following table, material data is given for standard pressure of 0.1 MPa (equivalent to 1 bar). Up to 99.63 °C (the boiling point of water), at this pressure water exists as a liquid. Above that, it exists as water vapor.
| Water/Steam Data Table at Standard Pressure (0.1 MPa) | ||||||||||
| T °C | V dm³/kg |
H kJ/kg |
U kJ/kg |
S kJ/(kg·K) |
cp kJ/(kg·K) |
γ 10–3/K |
λ mW / (m·K) |
η μPa·s |
σ1 mN/m | |
| 0 | lq | 1.0002 | 0.06 | -0.04 | -0.0001 | 4.228 | -0.080 | 561.0 | 1792 | 75.65 |
| 5 | 1.0000 | 21.1 | 21.0 | 0.076 | 4.200 | 0.011 | 570.6 | 1518 | 74.95 | |
| 10 | 1.0003 | 42.1 | 42.0 | 0.151 | 4.188 | 0.087 | 580.0 | 1306 | 74.22 | |
| 15 | 1.0009 | 63.0 | 62.9 | 0.224 | 4.184 | 0.152 | 589.4 | 1137 | 73.49 | |
| 20 | 1.0018 | 83.9 | 83.8 | 0.296 | 4.183 | 0.209 | 598.4 | 1001 | 72.74 | |
| 25 | 1.0029 | 104.8 | 104.7 | 0.367 | 4.183 | 0.259 | 607.2 | 890.4 | 71.98 | |
| 30 | 1.0044 | 125.8 | 125.7 | 0.437 | 4.183 | 0.305 | 615.5 | 797.7 | 71.20 | |
| 35 | 1.0060 | 146.7 | 146.6 | 0.505 | 4.183 | 0.347 | 623.3 | 719.6 | 70.41 | |
| 40 | 1.0079 | 167.6 | 167.5 | 0.572 | 4.182 | 0.386 | 630.6 | 653.3 | 69.60 | |
| 45 | 1.0099 | 188.5 | 188.4 | 0.638 | 4.182 | 0.423 | 637.3 | 596.3 | 68.78 | |
| 50 | 1.0121 | 209.4 | 209.3 | 0.704 | 4.181 | 0.457 | 643.6 | 547.1 | 67.95 | |
| 60 | 1.0171 | 251.2 | 251.1 | 0.831 | 4.183 | 0.522 | 654.4 | 466.6 | 66.24 | |
| 70 | 1.0227 | 293.1 | 293.0 | 0.955 | 4.187 | 0.583 | 663.1 | 404.1 | 64.49 | |
| 80 | 1.0290 | 335.0 | 334.9 | 1.075 | 4.194 | 0.640 | 670.0 | 354.5 | 62.68 | |
| 90 | 1.0359 | 377.0 | 376.9 | 1.193 | 4.204 | 0.696 | 675.3 | 314.6 | 60.82 | |
| 99.63 | lq | 1.0431 | 417.5 | 417.4 | 1.303 | 4.217 | 0.748 | 679.0 | 283.0 | 58.99 |
| g | 1694.3 | 2675 | 2505 | 7.359 | 2.043 | 2.885 | 25.05 | 12.26 | – | |
| 100 | g | 1696.1 | 2675 | 2506 | 7.361 | 2.042 | 2.881 | 25.08 | 12.27 | 58.92 |
| 200 | 2172.3 | 2874 | 2657 | 7.833 | 1.975 | 2.100 | 33.28 | 16.18 | 37.68 | |
| 300 | 2638.8 | 3073 | 2810 | 8.215 | 2.013 | 1.761 | 43.42 | 20.29 | 14.37 | |
| 500 | 3565.5 | 3488 | 3131 | 8.834 | 2.135 | 1.297 | 66.970 | 28.57 | – | |
| 750 | 4721.0 | 4043 | 3571 | 9.455 | 2.308 | 0.978 | 100.30 | 38.48 | – | |
| 1000 | 5875.5 | 4642 | 4054 | 9.978 | 2.478 | 0.786 | 136.3 | 47.66 | – | |
| 1 The values for surface tension for the liquid section of the table are for a liquid/air interface. Values for the gas section of the table are for a liquid/saturated steam interface. | ||||||||||
Triple Point
In the following table, material data is given with a pressure of 0.0006117 MPa (equivalent to 0.006117 bar). Up to a temperature of 0.01 °C, the triple point of water, water normally exists as ice, except for supercooled water, for which one data point is tabulated here. At the triple point ice can exist together with both liquid water and vapor. At higher temperatures the data is for water vapor only.
| Water/Steam Data Table at Triple Point Pressure (0.0006117 MPa) | |||||||||
| T °C | V dm³/kg |
H kJ/kg |
U kJ/kg |
S kJ/(kg·K) |
cp kJ/(kg·K) |
γ 10–3/K |
λ mW / (m·K) |
η μPa·s | |
| 0 | lq | 1.0002 | –0.04 | –0.04 | –0.0002 | 4.339 | –0.081 | 561.0 | 1792 |
| 0.1 | s | 1.0908 | –333.4 | –333.4 | –1.221 | 1.93 | 0.1 | 2.2 | – |
| lq | 1.0002 | 0.0 | 0 | 0 | 4.229 | –0.080 | 561.0 | 1791 | |
| g | 205986 | 2500 | 2374 | 9.154 | 1.868 | 3.672 | 17.07 | 9.22 | |
| 5 | g | 209913 | 2509 | 2381 | 9.188 | 1.867 | 3.605 | 17.33 | 9.34 |
| 10 | 213695 | 2519 | 2388 | 9.222 | 1.867 | 3.540 | 17.60 | 9.46 | |
| 15 | 217477 | 2528 | 2395 | 9.254 | 1.868 | 3.478 | 17.88 | 9.59 | |
| 20 | 221258 | 2537 | 2402 | 9.286 | 1.868 | 3.417 | 18.17 | 9.73 | |
| 25 | 225039 | 2547 | 2409 | 9.318 | 1.869 | 3.359 | 18.47 | 9.87 | |
| 30 | 228819 | 2556 | 2416 | 9.349 | 1.869 | 3.304 | 18.78 | 10.02 | |
| 35 | 232598 | 2565 | 2423 | 9.380 | 1.870 | 3.249 | 19.10 | 10.17 | |
| 40 | 236377 | 2575 | 2430 | 9.410 | 1.871 | 3.197 | 19.43 | 10.32 | |
| 45 | 240155 | 2584 | 2437 | 9.439 | 1.872 | 3.147 | 19.77 | 10.47 | |
| 50 | 243933 | 2593 | 2444 | 9.469 | 1.874 | 3.098 | 20.11 | 10.63 | |
| 60 | 251489 | 2612 | 2459 | 9.526 | 1.876 | 3.004 | 20.82 | 10.96 | |
| 70 | 259043 | 2631 | 2473 | 9.581 | 1.880 | 2.916 | 21.56 | 11.29 | |
| 80 | 266597 | 2650 | 2487 | 9.635 | 1.883 | 2.833 | 22.31 | 11.64 | |
| 90 | 274150 | 2669 | 2501 | 9.688 | 1.887 | 2.755 | 23.10 | 11.99 | |
| 100 | 281703 | 2688 | 2515 | 9.739 | 1.891 | 2.681 | 23.90 | 12.53 | |
| 200 | 357216 | 2879 | 2661 | 10.194 | 1.940 | 2.114 | 32.89 | 16.21 | |
| 300 | 432721 | 3076 | 2811 | 10.571 | 2.000 | 1.745 | 43.26 | 20.30 | |
| 500 | 583725 | 3489 | 3132 | 11.188 | 2.131 | 1.293 | 66.90 | 28.57 | |
| 750 | 772477 | 4043 | 3571 | 11.808 | 2.307 | 0.977 | 100.20 | 38.47 | |
| 1000 | 961227 | 4642 | 4054 | 12.331 | 2.478 | 0.785 | 136.30 | 47.66 | |
Saturated Vapor Pressure
The following table is based on different, complementary sources and approximation formulas, whose values are of various quality and accuracy. The values in the temperature range of –100 °C to 100 °C were inferred from D. Sunday (1982) and are quite uniform and exact. The values in the temperature range of the boiling point of the water up to the critical point (100 °C to 374 °C), are drawn from different sources and are substantially less accurate, hence they should be understood and used also only as approximate values.[13][14][15][16]
To use the values corrctly, consider the following points:
- The values apply only to smooth interfaces and in the absence other gases or gas mixtures such as air. Hence they apply only to pure phases and need a correction factor for systems in which air is present.
- The values were not computed according formulas widely used in the US, but using somewhat more exact formulas (see below), which can also also be used to compute further values in the appropriate temperature ranges.
- The saturated steam pressure over water in the temperature range of –100 °C to –50 °C is only extrapolated.
- The values have various units (Pa, hPa or bar), which must be considered when reading them.
Formulas
The table values for –100 °C to 100 °C were computed by the following formulas, where T is in Kelvins and vapor pressures, Pw and Pi are in Pa.
Over Liquid Water
- loge(Pw) = –6094.4642 T–1 + 21.1249952 – 2.724552×10–2 T + 1.6853396×10–5 T2 + 2.4575506 loge(T)
For Temperature Range: 173.15 K to 373.15 K or equivalently –100 °C to 100 °C
Over Ice
- loge(Pi) = –5504.4088 T–1 – 3.5704628 – 1.7337458×10–2 T + 6.5204209×10–6 T2 + 6.1295027 loge(T)
For temperature range: 173.15 K to 273.15 K or equivalently –100 °C to 0 °C
Triple point
An important basic value, which is not registered in the table, is the saturated vapor pressure at the triple point of water. The internationally accepted value according to measurements of Guildner, Johnson and Jones (1976) amounts to:
| Values of Saturated Vapor Pressure of Water | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Temp. t in °C |
Pi(t) over ice in Pa |
Pw(t) over water in Pa |
Temp. t in °C |
Pw(t) over water in hPa |
Temp. t in °C |
P(t) in bar |
Temp. t in °C |
P(t) in bar |
Temp. t in °C |
P(t) in bar |
| -100 | 0.0013957 | 0.0036309 | 0 | 6.11213 | 100 | 1.01 | 200 | 15.55 | 300 | 85.88 |
| -99 | 0.0017094 | 0.0044121 | 1 | 6.57069 | 101 | 1.05 | 201 | 15.88 | 301 | 87.09 |
| -98 | 0.0020889 | 0.0053487 | 2 | 7.05949 | 102 | 1.09 | 202 | 16.21 | 302 | 88.32 |
| -97 | 0.002547 | 0.0064692 | 3 | 7.58023 | 103 | 1.13 | 203 | 16.55 | 303 | 89.57 |
| -96 | 0.0030987 | 0.0078067 | 4 | 8.13467 | 104 | 1.17 | 204 | 16.89 | 304 | 90.82 |
| -95 | 0.0037617 | 0.0093996 | 5 | 8.72469 | 105 | 1.21 | 205 | 17.24 | 305 | 92.09 |
| -94 | 0.0045569 | 0.011293 | 6 | 9.35222 | 106 | 1.25 | 206 | 17.60 | 306 | 93.38 |
| -93 | 0.0055087 | 0.013538 | 7 | 10.0193 | 107 | 1.30 | 207 | 17.96 | 307 | 94.67 |
| -92 | 0.0066455 | 0.016195 | 8 | 10.728 | 108 | 1.34 | 208 | 18.32 | 308 | 95.98 |
| -91 | 0.0080008 | 0.019333 | 9 | 11.4806 | 109 | 1.39 | 209 | 18.70 | 309 | 97.31 |
| -90 | 0.0096132 | 0.023031 | 10 | 12.2794 | 110 | 1.43 | 210 | 19.07 | 310 | 98.65 |
| -89 | 0.011528 | 0.027381 | 11 | 13.1267 | 111 | 1.48 | 211 | 19.46 | 311 | 100 |
| -88 | 0.013797 | 0.032489 | 12 | 14.0251 | 112 | 1.53 | 212 | 19.85 | 312 | 101.37 |
| -87 | 0.016482 | 0.038474 | 13 | 14.9772 | 113 | 1.58 | 213 | 20.25 | 313 | 102.75 |
| -86 | 0.019653 | 0.045473 | 14 | 15.9856 | 114 | 1.64 | 214 | 20.65 | 314 | 104.15 |
| -85 | 0.02339 | 0.053645 | 15 | 17.0532 | 115 | 1.69 | 215 | 21.06 | 315 | 105.56 |
| -84 | 0.027788 | 0.063166 | 16 | 18.1829 | 116 | 1.75 | 216 | 21.47 | 316 | 106.98 |
| -83 | 0.032954 | 0.074241 | 17 | 19.3778 | 117 | 1.81 | 217 | 21.89 | 317 | 108.43 |
| -82 | 0.039011 | 0.087101 | 18 | 20.6409 | 118 | 1.86 | 218 | 22.32 | 318 | 109.88 |
| -81 | 0.046102 | 0.10201 | 19 | 21.9757 | 119 | 1.93 | 219 | 22.75 | 319 | 111.35 |
| -80 | 0.054388 | 0.11925 | 20 | 23.3854 | 120 | 1.99 | 220 | 23.19 | 320 | 112.84 |
| -79 | 0.064057 | 0.13918 | 21 | 24.8737 | 121 | 2.05 | 221 | 23.64 | 321 | 114.34 |
| -78 | 0.07532 | 0.16215 | 22 | 26.4442 | 122 | 2.12 | 222 | 24.09 | 322 | 115.86 |
| -77 | 0.088419 | 0.1886 | 23 | 28.1006 | 123 | 2.18 | 223 | 24.55 | 323 | 117.39 |
| -76 | 0.10363 | 0.21901 | 24 | 29.847 | 124 | 2.25 | 224 | 25.02 | 324 | 118.94 |
| -75 | 0.12127 | 0.25391 | 25 | 31.6874 | 125 | 2.32 | 225 | 25.49 | 325 | 120.51 |
| -74 | 0.14168 | 0.29390 | 26 | 33.6260 | 126 | 2.4 | 226 | 25.98 | 326 | 122.09 |
| -73 | 0.16528 | 0.33966 | 27 | 35.6671 | 127 | 2.47 | 227 | 26.46 | 327 | 123.68 |
| -72 | 0.19252 | 0.39193 | 28 | 37.8154 | 128 | 2.55 | 228 | 26.96 | 328 | 125.30 |
| -71 | 0.22391 | 0.45156 | 29 | 40.0754 | 129 | 2.62 | 229 | 27.46 | 329 | 126.93 |
| -70 | 0.26004 | 0.51948 | 30 | 42.452 | 130 | 2.7 | 230 | 27.97 | 330 | 128.58 |
| -69 | 0.30156 | 0.59672 | 31 | 44.9502 | 131 | 2.78 | 231 | 28.48 | 331 | 130.24 |
| -68 | 0.34921 | 0.68446 | 32 | 47.5752 | 132 | 2.87 | 232 | 29.01 | 332 | 131.92 |
| -67 | 0.40383 | 0.78397 | 33 | 50.3322 | 133 | 2.95 | 233 | 29.54 | 333 | 133.62 |
| -66 | 0.46633 | 0.89668 | 34 | 53.2267 | 134 | 3.04 | 234 | 30.08 | 334 | 135.33 |
| -65 | 0.53778 | 1.0242 | 35 | 56.2645 | 135 | 3.13 | 235 | 30.62 | 335 | 137.07 |
| -64 | 0.61933 | 1.1682 | 36 | 59.4513 | 136 | 3.22 | 236 | 31.18 | 336 | 138.82 |
| -63 | 0.71231 | 1.3306 | 37 | 62.7933 | 137 | 3.32 | 237 | 31.74 | 337 | 140.59 |
| -62 | 0.81817 | 1.5136 | 38 | 66.2956 | 138 | 3.42 | 238 | 32.31 | 338 | 142.37 |
| -61 | 0.93854 | 1.71950 | 39 | 69.9675 | 139 | 3.51 | 239 | 32.88 | 339 | 144.18 |
| -60 | 1.0753 | 1.9509 | 40 | 73.8127 | 140 | 3.62 | 240 | 33.47 | 340 | 146.00 |
| -59 | 1.2303 | 2.2106 | 41 | 77.8319 | 141 | 3.72 | 241 | 34.06 | 341 | 147.84 |
| -58 | 1.4060 | 2.5018 | 42 | 82.0536 | 142 | 3.82 | 242 | 34.66 | 342 | 149.71 |
| -57 | 1.6049 | 2.8277 | 43 | 86.4633 | 143 | 3.93 | 243 | 35.27 | 343 | 151.58 |
| -56 | 1.8296 | 3.1922 | 44 | 91.0757 | 144 | 4.04 | 244 | 35.88 | 344 | 153.48 |
| -55 | 2.0833 | 3.5993 | 45 | 95.8984 | 145 | 4.16 | 245 | 36.51 | 345 | 155.40 |
| -54 | 2.3694 | 4.0535 | 46 | 100.939 | 146 | 4.27 | 246 | 37.14 | 346 | 157.34 |
| -53 | 2.6917 | 4.5597 | 47 | 106.206 | 147 | 4.39 | 247 | 37.78 | 347 | 159.30 |
| -52 | 3.0542 | 5.1231 | 48 | 111.708 | 148 | 4.51 | 248 | 38.43 | 348 | 161.28 |
| -51 | 3.4618 | 5.7496 | 49 | 117.452 | 149 | 4.64 | 249 | 39.09 | 349 | 163.27 |
| -50 | 3.9193 | 6.4454 | 50 | 123.4478 | 150 | 4.76 | 250 | 39.76 | 350 | 165.29 |
| -49 | 4.4324 | 7.2174 | 51 | 129.7042 | 151 | 4.89 | 251 | 40.44 | 351 | 167.33 |
| -48 | 5.0073 | 8.0729 | 52 | 136.2304 | 152 | 5.02 | 252 | 41.12 | 352 | 169.39 |
| -47 | 5.6506 | 9.0201 | 53 | 143.0357 | 153 | 5.16 | 253 | 41.81 | 353 | 171.47 |
| -46 | 6.3699 | 10.068 | 54 | 150.1298 | 154 | 5.29 | 254 | 42.52 | 354 | 173.58 |
| -45 | 7.1732 | 11.225 | 55 | 157.5226 | 155 | 5.43 | 255 | 43.23 | 355 | 175.70 |
| -44 | 8.0695 | 12.503 | 56 | 165.2243 | 156 | 5.58 | 256 | 43.95 | 356 | 177.85 |
| -43 | 9.0685 | 13.911 | 57 | 173.2451 | 157 | 5.72 | 257 | 44.68 | 357 | 180.02 |
| -42 | 10.181 | 15.463 | 58 | 181.5959 | 158 | 5.87 | 258 | 45.42 | 358 | 182.21 |
| -41 | 11.419 | 17.17 | 59 | 190.2874 | 159 | 6.03 | 259 | 46.16 | 359 | 184.43 |
| -40 | 12.794 | 19.048 | 60 | 199.3309 | 160 | 6.18 | 260 | 46.92 | 360 | 186.66 |
| -39 | 14.321 | 21.11 | 61 | 208.7378 | 161 | 6.34 | 261 | 47.69 | 361 | 188.93 |
| -38 | 16.016 | 23.372 | 62 | 218.5198 | 162 | 6.50 | 262 | 48.46 | 362 | 191.21 |
| -37 | 17.893 | 25.853 | 63 | 228.6888 | 163 | 6.67 | 263 | 49.25 | 363 | 193.52 |
| -36 | 19.973 | 28.57 | 64 | 239.2572 | 164 | 6.84 | 264 | 50.05 | 364 | 195.86 |
| -35 | 22.273 | 31.544 | 65 | 250.2373 | 165 | 7.01 | 265 | 50.85 | 365 | 198.22 |
| -34 | 24.816 | 34.795 | 66 | 261.6421 | 166 | 7.18 | 266 | 51.67 | 366 | 200.61 |
| -33 | 27.624 | 38.347 | 67 | 273.4845 | 167 | 7.36 | 267 | 52.49 | 367 | 203.02 |
| -32 | 30.723 | 42.225 | 68 | 285.7781 | 168 | 7.55 | 268 | 53.33 | 368 | 205.47 |
| -31 | 34.140 | 46.453 | 69 | 298.5363 | 169 | 7.73 | 269 | 54.17 | 369 | 207.93 |
| -30 | 37.903 | 51.060 | 70 | 311.7731 | 170 | 7.92 | 270 | 55.03 | 370 | 210.43 |
| -29 | 42.046 | 56.077 | 71 | 325.5029 | 171 | 8.11 | 271 | 55.89 | 371 | 212.96 |
| -28 | 46.601 | 61.534 | 72 | 339.7401 | 172 | 8.31 | 272 | 56.77 | 372 | 215.53 |
| -27 | 51.607 | 67.466 | 73 | 354.4995 | 173 | 8.51 | 273 | 57.66 | 373 | 218.13 |
| -26 | 57.104 | 73.909 | 74 | 369.7963 | 174 | 8.72 | 274 | 58.56 | 374 | 220.64 |
| -25 | 63.134 | 80.902 | 75 | 385.6459 | 175 | 8.92 | 275 | 59.46 | 374.15 | 221.20 |
| -24 | 69.745 | 88.485 | 76 | 402.0641 | 176 | 9.14 | 276 | 60.38 | ||
| -23 | 76.987 | 96.701 | 77 | 419.0669 | 177 | 9.35 | 277 | 61.31 | ||
| -22 | 84.914 | 105.60 | 78 | 436.6708 | 178 | 9.57 | 278 | 62.25 | ||
| -21 | 93.584 | 115.22 | 79 | 454.8923 | 179 | 9.80 | 279 | 63.20 | ||
| -20 | 103.06 | 125.63 | 80 | 473.7485 | 180 | 10.03 | 280 | 64.17 | ||
| -19 | 113.41 | 136.88 | 81 | 493.2567 | 181 | 10.26 | 281 | 65.14 | ||
| -18 | 124.70 | 149.01 | 82 | 513.4345 | 182 | 10.50 | 282 | 66.12 | ||
| -17 | 137.02 | 162.11 | 83 | 534.3000 | 183 | 10.74 | 283 | 67.12 | ||
| -16 | 150.44 | 176.23 | 84 | 555.8714 | 184 | 10.98 | 284 | 68.13 | ||
| -15 | 165.06 | 191.44 | 85 | 578.1673 | 185 | 11.23 | 285 | 69.15 | ||
| -14 | 180.97 | 207.81 | 86 | 601.2068 | 186 | 11.49 | 286 | 70.18 | ||
| -13 | 198.27 | 225.43 | 87 | 625.009 | 187 | 11.75 | 287 | 71.22 | ||
| -12 | 217.07 | 244.37 | 88 | 649.5936 | 188 | 12.01 | 288 | 72.27 | ||
| -11 | 237.49 | 264.72 | 89 | 674.9806 | 189 | 12.28 | 289 | 73.34 | ||
| -10 | 259.66 | 286.57 | 90 | 701.1904 | 190 | 12.55 | 290 | 74.42 | ||
| -9 | 283.69 | 310.02 | 91 | 728.2434 | 191 | 12.83 | 291 | 75.51 | ||
| -8 | 309.75 | 335.16 | 92 | 756.1608 | 192 | 13.11 | 292 | 76.61 | ||
| -7 | 337.97 | 362.10 | 93 | 784.9639 | 193 | 13.40 | 293 | 77.72 | ||
| -6 | 368.52 | 390.95 | 94 | 814.6743 | 194 | 13.69 | 294 | 78.85 | ||
| -5 | 401.58 | 421.84 | 95 | 845.3141 | 195 | 13.99 | 295 | 79.99 | ||
| -4 | 437.31 | 454.88 | 96 | 876.9057 | 196 | 14.29 | 296 | 81.14 | ||
| -3 | 475.92 | 490.19 | 97 | 909.4718 | 197 | 14.60 | 297 | 82.31 | ||
| -2 | 517.62 | 527.93 | 98 | 943.0355 | 198 | 14.91 | 298 | 83.48 | ||
| -1 | 562.62 | 568.22 | 99 | 977.6203 | 199 | 15.22 | 299 | 84.67 | ||
| 0 | 611.153 | 611.213 | 100 | 1013.25 | 200 | 15.55 | 300 | 85.88 | ||
| Temp. t in °C |
Pi(t) over ice in Pa |
Pw(t) over water in Pa |
Temp. t in °C |
Pw(t) over water in hPa |
Temp. t in °C |
P(t) in bar |
Temp. t in °C |
P(t) in bar |
Temp. t in °C |
P(t) in barbar |
Material is copied in from
Drucktabellen
Die in der folgenden Tabelle dargestellten Größen sind temperatur- und teilweise auch druckabhängig, richten sich aber in jedem Fall nach dem Aggregatzustand des Wassers (hier s = fest; l = flüssig; g = gasförmig). Dieser wird durch Druck und Temperatur eindeutig bestimmt. Alle Daten wurden Grigull et. al. (1990) entnommen, welche sie nach der Vorgabe durch die "Formulation of the Thermodynamic Properties of Ordinary Water Substance for Scientific and General Use" (1984) der IAPWS mit einer verringerten Iterationsschranke berechneten. Es handelt sich um:
- - die Celsius-Temperatur in Grad Celsius
- v – das spezifische Volumen in Kubikdezimeter je Kilogramm
- h – die spezifische Enthalpie in Kilojoule je Kilogramm
- u – die spezifische Innere Energie in Kilojoule je Kilogramm
- s – die spezifische Entropie in Kilojoule je Kilogramm mal Kelvin
- cp - die spezifische Wärmekapazität bei konstantem Druck in Kilojoule je Kilogramm mal Kelvin
- γ – Volumenausdehnungskoeffzient in 10-3 durch Kelvin
- λ – Wärmeleitfähigkeit in Milliwatt je Meter mal Kelvin
- η – Viskosität in Mikropascal mal Sekunde
- σ – Oberflächenspannung in Millinewton je Meter
Standardbedingungen
In der folgenden Tabelle handelt es sich um die Stoffdaten bei Standarddruck (SATP), also 0,1 Megapascal (entspricht einem bar). Bis zu einer Temperatur von 99,63 °C, dem Siedepunkt des Wasser bei diesem Druck, liegt das Wasser als Flüssigkeit vor, darüber als Wasserdampf.
°C |
v dm³/kg |
h kJ/kg |
u kJ/kg |
s kJ/(kg·K) |
cp kJ/(kg·K) |
γ 10-3/K |
λ mW / (m·K) |
η μPa·s |
σ1 mN/m | |
| 0 | 1,0002 | 0,06 | -0,04 | -0,0001 | 4,228 | -0,080 | 561,0 | 1792 | 75,65 | |
| 5 | 1,0000 | 21,1 | 21,0 | 0,076 | 4,200 | 0,011 | 570,6 | 1518 | 74,95 | |
| 10 | 1,0003 | 42,1 | 42,0 | 0,151 | 4,188 | 0,087 | 580,0 | 1306 | 74,22 | |
| 15 | 1,0009 | 63,0 | 62,9 | 0,224 | 4,184 | 0,152 | 589,4 | 1137 | 73,49 | |
| 20 | 1,0018 | 83,9 | 83,8 | 0,296 | 4,183 | 0,209 | 598,4 | 1001 | 72,74 | |
| 25 | 1,0029 | 104,8 | 104,7 | 0,367 | 4,183 | 0,259 | 607,2 | 890,4 | 71,98 | |
| 30 | 1,0044 | 125,8 | 125,7 | 0,437 | 4,183 | 0,305 | 615,5 | 797,7 | 71,20 | |
| 35 | 1,0060 | 146,7 | 146,6 | 0,505 | 4,183 | 0,347 | 623,3 | 719,6 | 70,41 | |
| 40 | 1,0079 | 167,6 | 167,5 | 0,572 | 4,182 | 0,386 | 630,6 | 653,3 | 69,60 | |
| 45 | 1,0099 | 188,5 | 188,4 | 0,638 | 4,182 | 0,423 | 637,3 | 596,3 | 68,78 | |
| 50 | 1,0121 | 209,4 | 209,3 | 0,704 | 4,181 | 0,457 | 643,6 | 547,1 | 67,95 | |
| 60 | 1,0171 | 251,2 | 251,1 | 0,831 | 4,183 | 0,522 | 654,4 | 466,6 | 66,24 | |
| 70 | 1,0227 | 293,1 | 293,0 | 0,955 | 4,187 | 0,583 | 663,1 | 404,1 | 64,49 | |
| 80 | 1,0290 | 335,0 | 334,9 | 1,075 | 4,194 | 0,640 | 670,0 | 354,5 | 62,68 | |
| 90 | 1,0359 | 377,0 | 376,9 | 1,193 | 4,204 | 0,696 | 675,3 | 314,6 | 60,82 | |
| 99,63 | l | 1,0431 | 417,5 | 417,4 | 1,303 | 4,217 | 0,748 | 679,0 | 283,0 | 58,99 |
| g | 1694,3 | 2675 | 2505 | 7,359 | 2,043 | 2,885 | 25,05 | 12,26 | – | |
| 100 | 1696,1 | 2675 | 2506 | 7,361 | 2,042 | 2,881 | 25,08 | 12,27 | 58,92 | |
| 200 | 2172,3 | 2874 | 2657 | 7,833 | 1,975 | 2,100 | 33,28 | 16,18 | 37,68 | |
| 300 | 2638,8 | 3073 | 2810 | 8,215 | 2,013 | 1,761 | 43,42 | 20,29 | 14,37 | |
| 500 | 3565,5 | 3488 | 3131 | 8,834 | 2,135 | 1,297 | 66,970 | 28,57 | – | |
| 750 | 4721,0 | 4043 | 3571 | 9,455 | 2,308 | 0,978 | 100,30 | 38,48 | – | |
| 1000 | 5875,5 | 4642 | 4054 | 9,978 | 2,478 | 0,786 | 136,3 | 47,66 | – | |
| 1 Die Werte der Oberflächenspannung gelten nicht für den Normaldruck, sondern für den zur jeweiligen Temperatur gehörigen Sättigungsdampfdruck. | ||||||||||
Tripelpunkt
In der folgenden Tabelle handelt es sich um die Stoffdaten bei einem Druck von 0,0006117 Megapascal (entspricht 0,006117 bar). Bis zu einer Temperatur von 0,01 °C, dem Tripelpunkt des Wassers, liegt das Wasser normalerweise als Eis vor, wurde jedoch hier für unterkühltes Wasser tabelliert. Am Tripelpunkt selbst kann es sowohl als Eis als auch Flüssigkeit oder Wasserdampf vorliegen, bei höheren Temperaturen handelt es sich jedoch wiederum um Wasserdampf.
°C |
v dm³/kg |
h kJ/kg |
u kJ/kg |
s kJ/(kg·K) |
cp kJ/(kg·K) |
γ 10-3/K |
λ mW / (m·K) |
η μPa·s |
σ1 mN/m | |
| 0 | 1,0002 | -0,04 | -0,04 | -0,0002 | 4,339 | -0,081 | 561,0 | 1792 | – | |
| 0,1 | s | 1,0908 | -333,4 | -333,4 | -1,221 | 1,93 | 0,1 | 2,2 | – | – |
| l | 1,0002 | 0,0 | 0 | 0 | 4,229 | -0,080 | 561,0 | 1791 | – | |
| g | 205986 | 2500 | 2374 | 9,154 | 1,868 | 3,672 | 17,07 | 9,22 | – | |
| 5 | 209913 | 2509 | 2381 | 9,188 | 1,867 | 3,605 | 17,33 | 9,34 | – | |
| 10 | 213695 | 2519 | 2388 | 9,222 | 1,867 | 3,540 | 17,60 | 9,46 | – | |
| 15 | 217477 | 2528 | 2395 | 9,254 | 1,868 | 3,478 | 17,88 | 9,59 | – | |
| 20 | 221258 | 2537 | 2402 | 9,286 | 1,868 | 3,417 | 18,17 | 9,73 | – | |
| 25 | 225039 | 2547 | 2409 | 9,318 | 1,869 | 3,359 | 18,47 | 9,87 | – | |
| 30 | 228819 | 2556 | 2416 | 9,349 | 1,869 | 3,304 | 18,78 | 10,02 | – | |
| 35 | 232598 | 2565 | 2423 | 9,380 | 1,870 | 3,249 | 19,10 | 10,17 | – | |
| 40 | 236377 | 2575 | 2430 | 9,410 | 1,871 | 3,197 | 19,43 | 10,32 | – | |
| 45 | 240155 | 2584 | 2437 | 9,439 | 1,872 | 3,147 | 19,77 | 10,47 | – | |
| 50 | 243933 | 2593 | 2444 | 9,469 | 1,874 | 3,098 | 20,11 | 10,63 | – | |
| 60 | 251489 | 2612 | 2459 | 9,526 | 1,876 | 3,004 | 20,82 | 10,96 | – | |
| 70 | 259043 | 2631 | 2473 | 9,581 | 1,880 | 2,916 | 21,56 | 11,29 | – | |
| 80 | 266597 | 2650 | 2487 | 9,635 | 1,883 | 2,833 | 22,31 | 11,64 | – | |
| 90 | 274150 | 2669 | 2501 | 9,688 | 1,887 | 2,755 | 23,10 | 11,99 | – | |
| 100 | 281703 | 2688 | 2515 | 9,739 | 1,891 | 2,681 | 23,90 | 12,53 | – | |
| 200 | 357216 | 2879 | 2661 | 10,194 | 1,940 | 2,114 | 32,89 | 16,21 | – | |
| 300 | 432721 | 3076 | 2811 | 10,571 | 2,000 | 1,745 | 43,26 | 20,30 | – | |
| 500 | 583725 | 3489 | 3132 | 11,188 | 2,131 | 1,293 | 66,90 | 28,57 | – | |
| 750 | 772477 | 4043 | 3571 | 11,808 | 2,307 | 0,977 | 100,20 | 38,47 | – | |
| 1000 | 961227 | 4642 | 4054 | 12,331 | 2,478 | 0,785 | 136,30 | 47,66 | – | |
| 1 Die Werte der Oberflächenspannung sind hier identisch zur ersten Tabelle, wobei in gleicherweise der Sättigungsdampfdruck angewendet werden muss. | ||||||||||
Sättigungsdampfdruck
Folgende Tabelle basiert auf verschiedenen, sich gegenseitig ergänzenden Quellen bzw. Näherungsformeln, was jedoch auch nach sich zieht, dass die Werte von unterschiedlicher Güte und Genauigkeit sind. Die Werte des Temperaturbereichs von -100 °C bis 100 °C wurden aus D. Sonntag (1982) entnommen und sind daher recht einheitlich und genau, wenn auch nicht auf dem neuesten Stand. Die Werte des Temperaturbereichs vom Siedepunkt des Wassers bis zum kritischen Punkt, also von 100 °C bis 374 °C, stammen jedoch aus unterschiedlichen Quellen und sind daher wesentlich ungenauer, folglich sollten sie auch nur als Orientierungswerte verstanden und genutzt werden.
Zur richtigen Nutzung der Werte sind folgende Punkte zu beachten:
- Die Werte gelten nur für ebene Oberflächen und in der Abwesenheit anderer Gase bzw. Gasgemische wie Luft. Sie gelten also lediglich für reine Phasen und benötigen einen Korrekturfaktor bei der Anwesenheit von Luft.
- Die Werte wurden nicht nach der Magnus-Formel berechnet, sondern nach etwas genaueren Formeln (siehe unten), mit deren Hilfe sich auch weitere Werte in den entsprechenden Temperaturintervallen berechnen lassen.
- Die Sättigungsdampfdrücke über Wasser im Temperaturintervall von -100 °C bis -50 °C wurden lediglich extrapoliert.
- Die Werte haben unterschiedliche Einheiten (Pa, hPa oder bar), was es beim ablesen zu beachten gilt.
Formeln
Berechnet wurden die Tabellenwerte von -100 °C bis 100 °C durch folgende Formeln:
Über Wasser:
Temperaturintervall:
- , entspricht
Über Eis:
Temperaturintervall:
- , entspricht
Werden die Temperaturen in Kelvin eingesetzt, so ergibt sich der jeweilige Sättigungsdampfdruck E(T) in Pa.
Tripelpunkt
Ein wichtiger Grundwert, der nicht in die Tabelle eingetragen wurde, ist der Sättigungsdampfdruck beim Tripelpunkt des Wassers. Der international akzeptierte Bestwert nach Messungen von Guildner, Johnson und Jones (1976) beträgt:
Tabelle
| Temperatur t in °C |
(t) über Eis p in Pa |
(t) über Wasser p in Pa |
Temperatur t in °C |
E(t) über Wasser p in hPa |
Temperatur t in °C |
E(t) p in bar |
Temperatur t in °C |
E(t) p in bar |
Temperatur t in °C |
E(t) p in bar |
|---|---|---|---|---|---|---|---|---|---|---|
| -100 | 0,0013957 | 0,0036309 | 0 | 6,11213 | 100 | 1,01 | 200 | 15,55 | 300 | 85,88 |
| -99 | 0,0017094 | 0,0044121 | 1 | 6,57069 | 101 | 1,05 | 201 | 15,88 | 301 | 87,09 |
| -98 | 0,0020889 | 0,0053487 | 2 | 7,05949 | 102 | 1,09 | 202 | 16,21 | 302 | 88,32 |
| -97 | 0,002547 | 0,0064692 | 3 | 7,58023 | 103 | 1,13 | 203 | 16,55 | 303 | 89,57 |
| -96 | 0,0030987 | 0,0078067 | 4 | 8,13467 | 104 | 1,17 | 204 | 16,89 | 304 | 90,82 |
| -95 | 0,0037617 | 0,0093996 | 5 | 8,72469 | 105 | 1,21 | 205 | 17,24 | 305 | 92,09 |
| -94 | 0,0045569 | 0,011293 | 6 | 9,35222 | 106 | 1,25 | 206 | 17,6 | 306 | 93,38 |
| -93 | 0,0055087 | 0,013538 | 7 | 10,0193 | 107 | 1,3 | 207 | 17,96 | 307 | 94,67 |
| -92 | 0,0066455 | 0,016195 | 8 | 10,728 | 108 | 1,34 | 208 | 18,32 | 308 | 95,98 |
| -91 | 0,0080008 | 0,019333 | 9 | 11,4806 | 109 | 1,39 | 209 | 18,7 | 309 | 97,31 |
| -90 | 0,0096132 | 0,023031 | 10 | 12,2794 | 110 | 1,43 | 210 | 19,07 | 310 | 98,65 |
| -89 | 0,011528 | 0,027381 | 11 | 13,1267 | 111 | 1,48 | 211 | 19,46 | 311 | 100 |
| -88 | 0,013797 | 0,032489 | 12 | 14,0251 | 112 | 1,53 | 212 | 19,85 | 312 | 101,37 |
| -87 | 0,016482 | 0,038474 | 13 | 14,9772 | 113 | 1,58 | 213 | 20,25 | 313 | 102,75 |
| -86 | 0,019653 | 0,045473 | 14 | 15,9856 | 114 | 1,64 | 214 | 20,65 | 314 | 104,15 |
| -85 | 0,02339 | 0,053645 | 15 | 17,0532 | 115 | 1,69 | 215 | 21,06 | 315 | 105,56 |
| -84 | 0,027788 | 0,063166 | 16 | 18,1829 | 116 | 1,75 | 216 | 21,47 | 316 | 106,98 |
| -83 | 0,032954 | 0,074241 | 17 | 19,3778 | 117 | 1,81 | 217 | 21,89 | 317 | 108,43 |
| -82 | 0,039011 | 0,087101 | 18 | 20,6409 | 118 | 1,86 | 218 | 22,32 | 318 | 109,88 |
| -81 | 0,046102 | 0,10201 | 19 | 21,9757 | 119 | 1,93 | 219 | 22,75 | 319 | 111,35 |
| -80 | 0,054388 | 0,11925 | 20 | 23,3854 | 120 | 1,99 | 220 | 23,19 | 320 | 112,84 |
| -79 | 0,064057 | 0,13918 | 21 | 24,8737 | 121 | 2,05 | 221 | 23,64 | 321 | 114,34 |
| -78 | 0,07532 | 0,16215 | 22 | 26,4442 | 122 | 2,12 | 222 | 24,09 | 322 | 115,86 |
| -77 | 0,088419 | 0,1886 | 23 | 28,1006 | 123 | 2,18 | 223 | 24,55 | 323 | 117,39 |
| -76 | 0,10363 | 0,21901 | 24 | 29,847 | 124 | 2,25 | 224 | 25,02 | 324 | 118,94 |
| -75 | 0,12127 | 0,25391 | 25 | 31,6874 | 125 | 2,32 | 225 | 25,49 | 325 | 120,51 |
| -74 | 0,14168 | 0,2939 | 26 | 33,626 | 126 | 2,4 | 226 | 25,98 | 326 | 122,09 |
| -73 | 0,16528 | 0,33966 | 27 | 35,6671 | 127 | 2,47 | 227 | 26,46 | 327 | 123,68 |
| -72 | 0,19252 | 0,39193 | 28 | 37,8154 | 128 | 2,55 | 228 | 26,96 | 328 | 125,3 |
| -71 | 0,22391 | 0,45156 | 29 | 40,0754 | 129 | 2,62 | 229 | 27,46 | 329 | 126,93 |
| -70 | 0,26004 | 0,51948 | 30 | 42,452 | 130 | 2,7 | 230 | 27,97 | 330 | 128,58 |
| -69 | 0,30156 | 0,59672 | 31 | 44,9502 | 131 | 2,78 | 231 | 28,48 | 331 | 130,24 |
| -68 | 0,34921 | 0,68446 | 32 | 47,5752 | 132 | 2,87 | 232 | 29,01 | 332 | 131,92 |
| -67 | 0,40383 | 0,78397 | 33 | 50,3322 | 133 | 2,95 | 233 | 29,54 | 333 | 133,62 |
| -66 | 0,46633 | 0,89668 | 34 | 53,2267 | 134 | 3,04 | 234 | 30,08 | 334 | 135,33 |
| -65 | 0,53778 | 1,0242 | 35 | 56,2645 | 135 | 3,13 | 235 | 30,62 | 335 | 137,07 |
| -64 | 0,61933 | 1,1682 | 36 | 59,4513 | 136 | 3,22 | 236 | 31,18 | 336 | 138,82 |
| -63 | 0,71231 | 1,3306 | 37 | 62,7933 | 137 | 3,32 | 237 | 31,74 | 337 | 140,59 |
| -62 | 0,81817 | 1,5136 | 38 | 66,2956 | 138 | 3,42 | 238 | 32,31 | 338 | 142,37 |
| -61 | 0,93854 | 1,7195 | 39 | 69,9675 | 139 | 3,51 | 239 | 32,88 | 339 | 144,18 |
| -60 | 1,0753 | 1,9509 | 40 | 73,8127 | 140 | 3,62 | 240 | 33,47 | 340 | 146 |
| -59 | 1,2303 | 2,2106 | 41 | 77,8319 | 141 | 3,72 | 241 | 34,06 | 341 | 147,84 |
| -58 | 1,406 | 2,5018 | 42 | 82,0536 | 142 | 3,82 | 242 | 34,66 | 342 | 149,71 |
| -57 | 1,6049 | 2,8277 | 43 | 86,4633 | 143 | 3,93 | 243 | 35,27 | 343 | 151,58 |
| -56 | 1,8296 | 3,1922 | 44 | 91,0757 | 144 | 4,04 | 244 | 35,88 | 344 | 153,48 |
| -55 | 2,0833 | 3,5993 | 45 | 95,8984 | 145 | 4,16 | 245 | 36,51 | 345 | 155,4 |
| -54 | 2,3694 | 4,0535 | 46 | 100,939 | 146 | 4,27 | 246 | 37,14 | 346 | 157,34 |
| -53 | 2,6917 | 4,5597 | 47 | 106,206 | 147 | 4,39 | 247 | 37,78 | 347 | 159,3 |
| -52 | 3,0542 | 5,1231 | 48 | 111,708 | 148 | 4,51 | 248 | 38,43 | 348 | 161,28 |
| -51 | 3,4618 | 5,7496 | 49 | 117,452 | 149 | 4,64 | 249 | 39,09 | 349 | 163,27 |
| -50 | 3,9193 | 6,4454 | 50 | 123,4478 | 150 | 4,76 | 250 | 39,76 | 350 | 165,29 |
| -49 | 4,4324 | 7,2174 | 51 | 129,7042 | 151 | 4,89 | 251 | 40,44 | 351 | 167,33 |
| -48 | 5,0073 | 8,0729 | 52 | 136,2304 | 152 | 5,02 | 252 | 41,12 | 352 | 169,39 |
| -47 | 5,6506 | 9,0201 | 53 | 143,0357 | 153 | 5,16 | 253 | 41,81 | 353 | 171,47 |
| -46 | 6,3699 | 10,068 | 54 | 150,1298 | 154 | 5,29 | 254 | 42,52 | 354 | 173,58 |
| -45 | 7,1732 | 11,225 | 55 | 157,5226 | 155 | 5,43 | 255 | 43,23 | 355 | 175,7 |
| -44 | 8,0695 | 12,503 | 56 | 165,2243 | 156 | 5,58 | 256 | 43,95 | 356 | 177,85 |
| -43 | 9,0685 | 13,911 | 57 | 173,2451 | 157 | 5,72 | 257 | 44,68 | 357 | 180,02 |
| -42 | 10,181 | 15,463 | 58 | 181,5959 | 158 | 5,87 | 258 | 45,42 | 358 | 182,21 |
| -41 | 11,419 | 17,17 | 59 | 190,2874 | 159 | 6,03 | 259 | 46,16 | 359 | 184,43 |
| -40 | 12,794 | 19,048 | 60 | 199,3309 | 160 | 6,18 | 260 | 46,92 | 360 | 186,66 |
| -39 | 14,321 | 21,11 | 61 | 208,7378 | 161 | 6,34 | 261 | 47,69 | 361 | 188,93 |
| -38 | 16,016 | 23,372 | 62 | 218,5198 | 162 | 6,5 | 262 | 48,46 | 362 | 191,21 |
| -37 | 17,893 | 25,853 | 63 | 228,6888 | 163 | 6,67 | 263 | 49,25 | 363 | 193,52 |
| -36 | 19,973 | 28,57 | 64 | 239,2572 | 164 | 6,84 | 264 | 50,05 | 364 | 195,86 |
| -35 | 22,273 | 31,544 | 65 | 250,2373 | 165 | 7,01 | 265 | 50,85 | 365 | 198,22 |
| -34 | 24,816 | 34,795 | 66 | 261,6421 | 166 | 7,18 | 266 | 51,67 | 366 | 200,61 |
| -33 | 27,624 | 38,347 | 67 | 273,4845 | 167 | 7,36 | 267 | 52,49 | 367 | 203,02 |
| -32 | 30,723 | 42,225 | 68 | 285,7781 | 168 | 7,55 | 268 | 53,33 | 368 | 205,47 |
| -31 | 34,14 | 46,453 | 69 | 298,5363 | 169 | 7,73 | 269 | 54,17 | 369 | 207,93 |
| -30 | 37,903 | 51,06 | 70 | 311,7731 | 170 | 7,92 | 270 | 55,03 | 370 | 210,43 |
| -29 | 42,046 | 56,077 | 71 | 325,5029 | 171 | 8,11 | 271 | 55,89 | 371 | 212,96 |
| -28 | 46,601 | 61,534 | 72 | 339,7401 | 172 | 8,31 | 272 | 56,77 | 372 | 215,53 |
| -27 | 51,607 | 67,466 | 73 | 354,4995 | 173 | 8,51 | 273 | 57,66 | 373 | 218,13 |
| -26 | 57,104 | 73,909 | 74 | 369,7963 | 174 | 8,72 | 274 | 58,56 | 374 | 220,64 |
| -25 | 63,134 | 80,902 | 75 | 385,6459 | 175 | 8,92 | 275 | 59,46 | 374,15 | 221,2 |
| -24 | 69,745 | 88,485 | 76 | 402,0641 | 176 | 9,14 | 276 | 60,38 | ||
| -23 | 76,987 | 96,701 | 77 | 419,0669 | 177 | 9,35 | 277 | 61,31 | ||
| -22 | 84,914 | 105,6 | 78 | 436,6708 | 178 | 9,57 | 278 | 62,25 | ||
| -21 | 93,584 | 115,22 | 79 | 454,8923 | 179 | 9,8 | 279 | 63,2 | ||
| -20 | 103,06 | 125,63 | 80 | 473,7485 | 180 | 10,03 | 280 | 64,17 | ||
| -19 | 113,41 | 136,88 | 81 | 493,2567 | 181 | 10,26 | 281 | 65,14 | ||
| -18 | 124,7 | 149,01 | 82 | 513,4345 | 182 | 10,5 | 282 | 66,12 | ||
| -17 | 137,02 | 162,11 | 83 | 534,3 | 183 | 10,74 | 283 | 67,12 | ||
| -16 | 150,44 | 176,23 | 84 | 555,8714 | 184 | 10,98 | 284 | 68,13 | ||
| -15 | 165,06 | 191,44 | 85 | 578,1673 | 185 | 11,23 | 285 | 69,15 | ||
| -14 | 180,97 | 207,81 | 86 | 601,2068 | 186 | 11,49 | 286 | 70,18 | ||
| -13 | 198,27 | 225,43 | 87 | 625,009 | 187 | 11,75 | 287 | 71,22 | ||
| -12 | 217,07 | 244,37 | 88 | 649,5936 | 188 | 12,01 | 288 | 72,27 | ||
| -11 | 237,49 | 264,72 | 89 | 674,9806 | 189 | 12,28 | 289 | 73,34 | ||
| -10 | 259,66 | 286,57 | 90 | 701,1904 | 190 | 12,55 | 290 | 74,42 | ||
| -9 | 283,69 | 310,02 | 91 | 728,2434 | 191 | 12,83 | 291 | 75,51 | ||
| -8 | 309,75 | 335,16 | 92 | 756,1608 | 192 | 13,11 | 292 | 76,61 | ||
| -7 | 337,97 | 362,1 | 93 | 784,9639 | 193 | 13,4 | 293 | 77,72 | ||
| -6 | 368,52 | 390,95 | 94 | 814,6743 | 194 | 13,69 | 294 | 78,85 | ||
| -5 | 401,58 | 421,84 | 95 | 845,3141 | 195 | 13,99 | 295 | 79,99 | ||
| -4 | 437,31 | 454,88 | 96 | 876,9057 | 196 | 14,29 | 296 | 81,14 | ||
| -3 | 475,92 | 490,19 | 97 | 909,4718 | 197 | 14,6 | 297 | 82,31 | ||
| -2 | 517,62 | 527,93 | 98 | 943,0355 | 198 | 14,91 | 298 | 83,48 | ||
| -1 | 562,62 | 568,22 | 99 | 977,6203 | 199 | 15,22 | 299 | 84,67 | ||
| 0 | 611,153 | 611,213 | 100 | 1013,25 | 200 | 15,55 | 300 | 85,88 | ||
Literatur
- L.A. Guildner, D.P. Johnson und F.E. Jones (1976): Vapor pressure of Water at Its Triple Point. J. Res. NBS - A, Vol. 80A, No. 3, p. 505 - 521
- Klaus Scheffler (1981): Wasserdampftafeln: thermodynam. Eigenschaften von Wasser u. Wasserdampf bis 800°C u. 800 bar (Water Vapor Tables: Thermodynamic Characteristics of Water and Water Vapor to 800°C and 800 bar), Berlin [u.a.] ISBN 3540109307
- D. Sonntag und D. Heinze (1982): Sättigungsdampfdruck- und Sättigungsdampfdichtetafeln für Wasser und Eis. (Saturated Vapor Pressure and Saturated Vapor Density Tables for Water and Ice)(1. Aufl.), VEB Deutscher Verlag für Grundstoffindustrie
- Ulrich Grigull, Johannes Staub, Peter Schiebener (1990): Steam Tables in SI-Units - Wasserdampftafeln. Springer-Verlagdima gmbh
Original draft of Frederick Jacobi article
Frederick Jacobi (born May 4,1891 in San Francisco, California; died October 24, 1952 in New York City of heart failure) was a prolific American composer, whose works include symphonies, concerti, chamber music, works for solo piano and for solo organ, lieder, and one opera. Besides composing, his career included teaching at Juilliard School of Music and serving as the director of the American section of the International Society for Contemporary Music.
Early life
Frederick Jacobi was the son of San Francisco wholesale wine merchant, Frederick Jacobi Sr. and Flora Brandenstein, whom Frederick Sr. had married in 1875. During the composer's childhood years, he demonstrated his musical talent, composing short pieces at the piano and playing tunes from contemporary musical comedies by ear. In these years the family traveled each summer to visit relatives in New York City. The scenery of those cross-country train rides later provided the themes of a number of his nature-inspired compositions.
Musical Training and Career
When Frederick Sr. died in 1911, Frederick Jr. inherited the estate, which provided him enough wealth that he could devote his entire livelihood to music. In his twenties Jacobi studied music and composition under such masters as Isidore Philipp of the Paris Conservatory, Ernest Bloch and Rubin Goldmark in New York, and Paul Juon in Berlin.
In 1917, while working as a vocal coach and assistant conductor at the Metropolitan Opera, he met and married Irene Schwarcz, who, at the time, was studying piano at the New York Institute of Musical Art (which later became Juilliard). Irene would go on to become an accomplished concert pianist, and played piano parts in many performances and recordings of Jacobi's works.
Jacobi enlisted in the army shortly after marrying Irene, where he served as a saxaphone player in the Alcatraz Army Band. He was discharged in 1919, at which time he moved to New York to be in closer contact with the American composers of the time. For the remainder of his life he published and performed new works nearly every year -- sometimes several in the same year (see compositions section). Major American orchestras such as the New York Philharmonic, the Philadelphia Orchestra, and the Boston, Chicago, and San Francisco symphonies performed Jacobi's orchestral compositions during the years of his life.
In works from what has become known as Jacobi's Indian period (late 1920's and early 1930's), he incorporated rhythms and other elements from indigenous Native American music he had heard in his travels through the American southwest. Indeed he spent the winter of 1927 with the Navajo and Pueblo of New Mexico studying their music.
In 1942-1944 Jacobi collaborated with playwright and librettist, Herman Voaden, to produce the opera, The Prodigal Son, which debuted at the American Opera Society of Chicago in May of 1945.
Jacobi is also known as a composer of works with Judaic themes. His interest in this genre began with a 1930 commission from Temple Emanu-El of New York for a sabbath evening service. This soon led to other works with biblical titles that explored the musical traditions of Judaism. Although little of Jacobi's secular work is performed today, his liturgical works continue to enjoy performaces in synagogues.
Jacobi's work largely rejects the polytonality and atonality that was popular with the avante-garde composers of his time. Instead he finds his influence in the classical and romantic periods. New York Times critic Olin Downes described the aesthetics of Jacobi's music as "not so much of the 20th as of the 19th century."
Frederick Jacobi quotes on musical composition
"I am a great believer in melody; a believer, too, that music should give pleasure and not try to solve philosophical problems."
"The surest way to kill whatever originality one possesses within himself is to try to be original!"
Links
Compositions
- 1915 The Pied Piper, Symphonic Poem
- 1916 Three Songs to Poems by Sarojini Naidu (“The Faery Isle of Janjira,” “Love and Death,” “In the Night,” for high voice and piano)
- 1917 A California Suite (for orchestra)
- 1918 Nocturne, for string quartet
- 1918 Psalmody (piano vocal score)
- 1920 Three Songs, for high voice with piano (words by Sarojini Naidu; “Palanquin-Bearers,” “In a Time of Flowers,” “From a Latticed Balcony”)
- 1920 The Eve of Saint Agnes (25 min. Symphonic prelude after the poem of John Keats)
- 1921 Three Preludes for Violin and Piano
- 1921 Morning and Evening at Blue Hill (for two violins and string orchestra with piano)
- 1921 A Festival Prelude (for orchestra)
- 1922 Symphony No. 1 (Subtitled Assyrian, 22 min.)
- 1922 Three Songs to Poems by Chaucer (for voice and piano) “Roundel” and “Ballade” published as Two Poems by Geoffrey Chaucer
- 1923 Two Assyrian Prayers (piano vocal score)
- 1923 Two Assyrian Prayers (Soprano or Tenor and chamber orchestra, 12 min. French text by Rebecca Godchaux. “To Ishtar” and “To Bel-Marduk”)
- 1924 Three Preludes for Violin, with orchestral accompaniment
- 1924 String Quartet (Based) on (American) Indian Themes (18 min.)
- 1925 The Poet in the Desert (after the poem by C.E.S. Wood, for orchestra, chorus and baritone solo)
- 1926 Nocturne (for flute and small orchestra; 5 min.) Rewritten second movement of Symphony No. 1, 1922)
- 1926 Marsyas (for violin and piano)
- 1927-28 Indian Dances/Danses Indiennes/Indianische Tänze (16 ½ min.) ( Buffalo Dance, Butterfly Dance, War Dance, Corn Dance; Suite for Orchestra)
- 1930-31 Sabbath Evening Service According to the Union Prayer Book (Friday Evening Service, baritone solo/cantor, mixed chorus, a capella; 20 min.)
- 1932 Concerto (Three Psalms) for Cello and Orchestra (16 min.) Reduction for Piano and Cello,
- 1933 String Quartet No. 2 (23 min.)
- 1933 Six Pieces for the Organ for Use in the Synagogue. One piece published as Prelude.
- 1933 Three Preludes for Organ.
- 1934-35 Concerto for Piano and Orchestra (26 min.)
- 1934 Piano Pieces for Children (includes A Lovely Little Movie Actress, Once Upon a Time, A Charming Prince, There Was a Wicked Fairy and Six Caprices) A Lovely Little Movie Actress and Once Upon a Time published separately.
- 1936 Scherzo for Flute, Oboe, Clarinet, Bassoon and Horn (5 min.) (Scherzo for Wind Instruments)
- 1936-37 Concerto for Violin and Orchestra (16 min.)
- 1937 Cadenza to Mozart’s Rondo for Piano and Orchestra (Kochel No. 386)
- 1937 Swing Boy (violin and piano)
- 1938 Hagiographa: Three Biblical Narratives for String Quartet and Piano (26 min.)
- 1938 Preludes on Traditional Melodies
- 1939 Ave Rota: (Hail to the Wheel [of Fortune]) Three Pieces in Multiple Style for Small Orchestra and Piano (“The Swing” [“La Balançoire”], “The Merman” and “May-Dance;” written for the Juilliard Alumni). (14 min. The same for large orchestra and piano)
- 1939 Dunam Po (“A Dunam Here”) Palestinian folk song arrangement published in Hans Nathan, ed. Folk Songs of the New Palestine.
- 194? Variations on a Theme by Moussorgsky (for cello and piano)
- 1940 Shemesh (based on a Palestinian Folk Song) Cello and Piano
- 1940 Rhapsody for Harp and String Orchestra (8 min.)
- 1941 Fantasy for Viola and Piano (9 min.)
- 1941 Ode for Orchestra (12 min.)
- 1941 Cadenza for Mozart’s Concerto for Piano and Orchestra in C Minor (Kochel No. 491)
- 1941 Night Piece for Flute and Small Orchestra (5 min.) (Rewritten Nocturne in Niniveh, 1926)
- 1941 Night Piece and Dance, for flute and piano.
- 1942 Ballade for Violin and Piano (11 min.)
- 1942 Hymn for Men’s Chorus (text by Saadia Gaon; 5 min.)
- 1942 From the Prophet Nehemiah: Three Excerpts for Voice and Two Pianos (5, 4 and 6 minutes respectively)
- 1942-44 The Prodigal Son: Opera in Three Acts based on Four Early American Prints. Text by Herman Voaden. (Full orchestra, 2 ½ hours). [Act I of first version inscribed “ July 18, 1943, Riverdale, N.Y.”; Act II “ Nov. 17, 1943” and Act III “ Sept. 9, 1944”]
- 1943 Penelope (arrangement for viola and piano from the 1921 Vocalises.)
- 1944 Dances From The Prodigal Son Arranged for Two Pianos, Four Hands (10 min.) [polka, polonaise, waltz, tarantella]
- 1944 Night Piece for Flute, Oboe, Clarinet, String Quintet and Piano
- 1944 Music for Monticello (Trio for Flute, Cello and Piano, 20 min.)
- 1945 String Quartet No. 3 (26 min.)
- 1945 Ahavas Olom (Ahavat Olam; 3 min.) (For tenor solo/cantor mixed voices and organ)
- 1945 Kaddish (for organ)
- 1945 Toccata (for organ)
- 1945 Toccata for piano solo. From Prelude and Toccata.
- 1945 Prelude in E Minor, for piano From Prelude and Toccata.
- 1945 Impressions from the Odyssey (three pieces for violin and piano; “Ulysses,” “Penelope,” “The Return”)
- 1945 Fantasy Sonata for Piano (9 min.)
- 1945 Four Dances From The Prodigal Son (orchestra, 18 min.)
- 1946 Concertino for Piano and String Orchestra (17 min.)
- 1946 Kaddish (for cantor, chorus and organ)
- 1946 Two Pieces in Sabbath Mood (Kaddish and Oneg Shabbat) (for orchestra, 2 min. and 9 min. Originally composed as two separate works for organ solo: Kaddish and Toccata; transcribed for small orchestra, 1946)
- 1946 Moods (for piano)
- 1946 Introduction and Toccata, for piano solo
- 1946 Prelude in E Minor for piano solo
- 1946 Contemplation (to a poem by William Blake, for mixed voices with piano accompaniment; 5:30 min.)
- 1946 Toccata (for organ)
- 1947 Symphony in C (Symphony No. 2, 21 min.)
- 1947 Meditation for Trombone and Piano
- 1947 Suite Fantasque (for piano)
- 1948 Three Songs to Words by Philip Freneau (for medium voice and piano). (“On the Sleep of Plants” [1790], “Elegy” [1786], “Ode to Freedom” [1795])
- 1948 Ode to Zion (text by Jehuda Halevi) for mixed voices and two harps
- 1948 Two Dances From The Prodigal Son (arranged for piano, four hands by the composer) [waltz, polka]
- 1948 Music Hall: Overture for Orchestra (6 min.)
- 1949 Yeibichai (Yébiché): Variations for Orchestra on an American Indian Theme (9 min.)
- 1949? Tuari: Nocturne for String Orchestra (“From the [Lento movement of] the String Quartet on Indian Themes”)
- 1949 Music Hall Suite
- 1949 Fanfare, in Memory of James Whitcomb Riley: Born 1849 (wind instruments and percussion)
- 1949 Ashrey Haish (arrangement for mixed voices and string orchestra of a Zionist song by Mordecai Zaira)
- 1950 Three Quiet Preludes (for organ)
- 1950 Ballade Concertante for two pianos
- 1950 Ballade Concertante (Symphonie Concertante) for piano and orchestra
- 1950-51 Sonata for Cello and Piano
- 1951 Two Pieces for Flute and Orchestra: Night Piece and Dance (Nocturne in Nineveh and Dance
- 1951 Capriccio for Violin and Piano
- 1951 Violin Pieces (with piano; “Alpha,” “Ad Astram,” “Bärentanz”)
- 1951 Night Piece and Dancefor Flute and Piano (Nocturne in Niniveh, for flute and piano)
- 1951-52 Arvit L’Shabbat (Friday Evening Service No. 2) for organ, baritone solo/cantor, mixed voices
- 1952 O May the Words for organ and mixed voices
- 1952 Serenade (Revised Ballade/Symphonie Concertante; arrangement for two pianos by the composer)
- 1952 Serenade for Piano and Orchestra (Revised Ballade/Symphonie Concertante)
Density of ethanol at various temperatures
Data obtained from Lange's Handbook of Chemistry, 10th ed.
| Temp. | Density | Temp. | Density | Temp. | Density | ||
| 0°C | 0.80625 | 13°C | 0.79535 | 26°C | 0.78437 | ||
| 1°C | 0.80541 | 14°C | 0.79451 | 27°C | 0.78352 | ||
| 2°C | 0.80457 | 15°C | 0.79367 | 28°C | 0.78267 | ||
| 3°C | 0.80374 | 16°C | 0.79283 | 29°C | 0.78182 | ||
| 4°C | 0.80290 | 17°C | 0.79198 | 30°C | 0.78097 | ||
| 5°C | 0.80207 | 18°C | 0.79114 | 31°C | 0.78012 | ||
| 6°C | 0.80123 | 19°C | 0.79029 | 32°C | 0.77927 | ||
| 7°C | 0.80039 | 20°C | 0.78945 | 33°C | 0.77841 | ||
| 8°C | 0.79956 | 21°C | 0.78860 | 34°C | 0.77756 | ||
| 9°C | 0.79872 | 22°C | 0.78775 | 35°C | 0.77671 | ||
| 10°C | 0.79788 | 23°C | 0.78691 | 36°C | 0.77585 | ||
| 11°C | 0.79704 | 24°C | 0.78606 | 37°C | 0.77500 | ||
| 12°C | 0.79620 | 25°C | 0.78522 | 38°C | 0.77414 | ||
| 39°C | 0.77329 |
Properties of aqueous ethanol solutions
Data obtained from Lange's Handbook of Chemistry, 10th ed. The annotation, d a°C/b°C, indicates density of solution at temperature a divided by density of pure water at temperature b.
| % wt ethanol | % vol ethanol | grams ethanol per 100 cc 15.56°C | d 10°C/4°C | d 20°C/4°C | d 25°C/4°C | d 30°C/4°C | d 20°C/20°C | d 25°C/25°C | freezing temp. |
| 0.0 | 0.0 | 0.0 | 0.99973 | 0.99823 | 0.99708 | 0.99568 | 1.00000 | 1.00000 | 0°C |
| 1.0 | 0.99785 | 0.99636 | 0.99520 | 0.99379 | 0.99813 | 0.99811 | |||
| 2.0 | 0.99602 | 0.99453 | 0.99336 | 0.99194 | 0.99629 | 0.99627 | |||
| 2.5 | 3.13 | 0.99363 | –1°C | ||||||
| 3.0 | 0.99426 | 0.99275 | 0.99157 | 0.99014 | 0.99451 | 0.99447 | |||
| 4.0 | 5.00 | 3.97 | 0.99258 | 0.99103 | 0.98984 | 0.98839 | 0.99279 | 0.99274 | |
| 4.8 | 6.00 | 4.76 | 0.98971 | –2°C | |||||
| 5.0 | 0.99098 | 0.98938 | 0.98817 | 0.98670 | 0.99113 | 0.99106 | |||
| 5.05 | 6.30 | 5.00 | 0.98930 | ||||||
| 6.0 | 0.98946 | 0.98780 | 0.98656 | 0.98507 | 0.98955 | 0.98945 | |||
| 6.8 | 8.47 | 0.98658 | –3°C | ||||||
| 7.0 | 0.98801 | 0.98627 | 0.98500 | 0.98347 | 0.98802 | 0.98788 | |||
| 8.0 | 0.98660 | 0.98478 | 0.98346 | 0.98189 | 0.98653 | 0.98634 | |||
| 9.0 | 0.98524 | 0.98331 | 0.98193 | 0.98031 | 0.98505 | 0.98481 | |||
| 10.0 | 12.40 | 9.84 | 0.98393 | 0.98187 | 0.98043 | 0.97575 | 0.98361 | 0.98330 | |
| 11.0 | 0.98267 | 0.98047 | 0.97897 | 0.97723 | 0.98221 | 0.98184 | |||
| 11.3 | 14.0 | 11.11 | 0.98006 | –5°C | |||||
| 12.0 | 0.98145 | 0.97910 | 0.97753 | 0.97573 | 0.98084 | 0.98039 | |||
| 13.0 | 0.98026 | 0.97775 | 0.97611 | 0.97424 | 0.97948 | 0.97897 | |||
| 13.78 | 17.00 | 13.49 | 0.98658 | –6.1°C | |||||
| 14.0 | 0.97911 | 0.97643 | 0.97472 | 0.97278 | 0.97816 | 0.97757 | |||
| 15.0 | 0.97800 | 0.97514 | 0.97334 | 0.97133 | 0.97687 | 0.97619 | |||
| 15.02 | 18.50 | 14.68 | 0.97511 | ||||||
| 16.0 | 0.97692 | 0.97387 | 0.97199 | 0.96990 | 0.97560 | 0.97484 | |||
| 16.4 | 20.2 | 0.97336 | –7.5°C | ||||||
| 17.0 | 0.97583 | 0.97259 | 0.97062 | 0.96844 | 0.97431 | 0.97346 | |||
| 17.5 | 21.5 | 0.97194 | –8.7°C | ||||||
| 18.0 | 22.10 | 17.54 | 0.97473 | 0.97129 | 0.96923 | 0.96697 | 0.97301 | 0.97207 | |
| 18.8 | 23.1 | 0.97024 | –9.4°C | ||||||
| 19.0 | 0.97363 | 0.96997 | 0.96782 | 0.96547 | 0.97169 | 0.97065 | |||
| 20.0 | 0.97252 | 0.96864 | 0.96639 | 0.96395 | 0.97036 | 0.96922 | |||
| 20.01 | 24.50 | 19.44 | 0.96863 | ||||||
| 20.3 | 24.8 | 0.96823 | –10.6°C | ||||||
| 21.0 | 0.97139 | 0.96729 | 0.96495 | 0.96242 | 0.96901 | 0.96778 | |||
| 22.0 | 0.97024 | 0.96592 | 0.96348 | 0.96087 | 0.96763 | 0.96630 | |||
| 22.11 | 27.00 | 21.43 | 0.96578 | –12.2°C | |||||
| 23.0 | 0.96907 | 0.96453 | 0.96199 | 0.95929 | 0.96624 | 0.96481 | |||
| 24.0 | 0.96787 | 0.96312 | 0.96048 | 0.95769 | 0.96483 | 0.96329 | |||
| 24.2 | 29.5 | 0.96283 | –14.0°C | ||||||
| 25.0 | 30.40 | 24.12 | 0.96665 | 0.96168 | 0.95895 | 0.95607 | 0.96339 | 0.96176 | |
| 26.0 | 0.96539 | 0.96020 | 0.95738 | 0.95422 | 0.96190 | 0.96018 | |||
| 26.7 | 32.4 | 0.95914 | –16.0°C | ||||||
| 27.0 | 0.96406 | 0.95867 | 0.95576 | 0.95272 | 0.96037 | 0.95856 | |||
| 28.0 | 33.90 | 26.90 | 0.96268 | 0.95710 | 0.95410 | 0.95098 | 0.95880 | 0.95689 | |
| 29.0 | 0.96125 | 0.95548 | 0.95241 | 0.94922 | 0.95717 | 0.95520 | |||
| 29.9 | 36.1 | 0.95400 | –18.9°C | ||||||
| 30.0 | 36.20 | 28.73 | 0.95977 | 0.95382 | 0.95067 | 0.94741 | 0.95551 | 0.95345 | |
| 31.0 | 0.95823 | 0.95212 | 0.94890 | 0.94557 | 0.95381 | 0.95168 | |||
| 32.0 | 0.95665 | 0.95038 | 0.94709 | 0.94370 | 0.95207 | 0.94986 | |||
| 33.0 | 0.95502 | 0.94860 | 0.94525 | 0.94180 | 0.95028 | 0.94802 | |||
| 33.8 | 40.5 | 0.94715 | –23.6°C | ||||||
| 34.0 | 0.95334 | 0.94679 | 0.94337 | 0.93986 | 0.94847 | 0.94613 | |||
| 35.0 | 0.95162 | 0.94494 | 0.94146 | 0.93790 | 0.94662 | 0.94422 | |||
| 35.04 | 41.90 | 33.25 | 0.94486 | ||||||
| 36.0 | 0.94986 | 0.94306 | 0.93952 | 0.93591 | 0.94473 | 0.94227 | |||
| 37.0 | 0.94805 | 0.94114 | 0.93756 | 0.93390 | 0.94281 | 0.94031 | |||
| 38.0 | 0.94620 | 0.93919 | 0.93556 | 0.93186 | 0.94086 | 0.93830 | |||
| 39.0 | 46.3 | 0.94431 | 0.93720 | 0.93353 | 0.92979 | 0.93886 | 0.93626 | –28.7°C | |
| 40.0 | 0.94238 | 0.93518 | 0.93148 | 0.92770 | 0.93684 | 0.93421 | |||
| 40.04 | 47.40 | 37.61 | 0.93510 | ||||||
| 41.0 | 0.94042 | 0.93314 | 0.92940 | 0.92558 | 0.93479 | 0.93212 | |||
| 42.0 | 0.93842 | 0.93107 | 0.92729 | 0.92344 | 0.93272 | 0.93001 | |||
| 43.0 | 0.93639 | 0.92897 | 0.92516 | 0.92128 | 0.93062 | 0.92787 | |||
| 44.0 | 0.93433 | 0.92685 | 0.92301 | 0.91910 | 0.92849 | 0.92571 | |||
| 45.0 | 0.93226 | 0.92472 | 0.92085 | 0.91692 | 0.92636 | 0.92355 | |||
| 45.31 | 53.00 | 42.07 | 0.92406 | ||||||
| 46.0 | 0.93017 | 0.92257 | 0.91868 | 0.91472 | 0.92421 | 0.92137 | |||
| 46.3 | 53.8 | 0.92193 | –33.9°C | ||||||
| 47.0 | 0.92806 | 0.92041 | 0.91649 | 0.91250 | 0.92204 | 0.91917 | |||
| 48.0 | 0.92593 | 0.91823 | 0.91429 | 0.91028 | 0.91986 | 0.91697 | |||
| 49.0 | 0.92379 | 0.91604 | 0.91208 | 0.90805 | 0.91766 | 0.91475 | |||
| 50.0 | 0.92162 | 0.91384 | 0.90985 | 0.90580 | 0.91546 | 0.91251 | |||
| 50.16 | 58.0 | 46.04 | 0.91349 | ||||||
| 51.0 | 0.91943 | 0.91160 | 0.90760 | 0.90353 | 0.91322 | 0.91026 | |||
| 52.0 | 0.91723 | 0.90936 | 0.90524 | 0.90125 | 0.91097 | 0.90799 | |||
| 53.0 | 0.91502 | 0.90711 | 0.90307 | 0.89896 | 0.90872 | 0.90571 | |||
| 54.0 | 0.91279 | 0.90485 | 0.90079 | 0.89667 | 0.90645 | 0.90343 | |||
| 55.0 | 0.91055 | 0.90258 | 0.89850 | 0.89437 | 0.90418 | 0.90113 | |||
| 55.16 | 63.0 | 50.00 | 0.90220 | ||||||
| 56.0 | 0.90831 | 0.90031 | 0.89621 | 0.89206 | 0.90191 | 0.89833 | |||
| 56.1 | 63.6 | 0.90008 | –41.0°C | ||||||
| 57.0 | 0.90607 | 0.89803 | 0.89392 | 0.88975 | 0.89962 | 0.89654 | |||
| 58.0 | 0.90381 | 0.89574 | 0.89162 | 0.88744 | 0.89733 | 0.89423 | |||
| 59.0 | 0.90154 | 0.89344 | 0.88931 | 0.88512 | 0.89502 | 0.89191 | |||
| 60.0 | 0.89927 | 0.89113 | 0.88699 | 0.88278 | 0.89271 | 0.88959 | |||
| 60.33 | 68.0 | 53.98 | 0.89038 | ||||||
| 61.0 | 0.89898 | 0.88882 | 0.88466 | 0.88044 | 0.89040 | 0.88725 | |||
| 62.0 | 0.89468 | 0.88650 | 0.88233 | 0.87809 | 0.88807 | 0.88491 | |||
| 63.0 | 0.89237 | 0.88417 | 0.87998 | 0.87574 | 0.88574 | 0.88256 | |||
| 64.0 | 0.89006 | 0.88183 | 0.87763 | 0.87337 | 0.88339 | 0.88020 | |||
| 65.0 | 0.88774 | 0.87948 | 0.87527 | 0.87100 | 0.88104 | 0.87783 | |||
| 66.0 | 0.88541 | 0.87713 | 0.87291 | 0.86863 | 0.87869 | 0.87547 | |||
| 67.0 | 0.88308 | 0.87477 | 0.87054 | 0.86625 | 0.87632 | 0.87309 | |||
| 68.0 | 0.88071 | 0.87241 | 0.86817 | 0.86387 | 0.87396 | 0.87071 | |||
| 69.0 | 0.87839 | 0.87004 | 0.86579 | 0.86148 | 0.87158 | 0.86833 | |||
| 70.0 | 0.87602 | 0.86766 | 0.86340 | 0.85908 | 0.86920 | 0.86593 | |||
| 71.0 | 0.87365 | 0.86527 | 0.86100 | 0.85667 | 0.86680 | 0.86352 | |||
| 71.9 | 78.3 | 0.86311 | –51.3°C | ||||||
| 72.0 | 0.87127 | 0.86287 | 0.85859 | 0.85426 | 0.86440 | 0.86110 | |||
| 73.0 | 0.86888 | 0.86047 | 0.85618 | 0.85184 | 0.86200 | 0.85869 | |||
| 74.0 | 0.86648 | 0.85806 | 0.85376 | 0.84941 | 0.85958 | 0.85626 | |||
| 75.0 | 0.86408 | 0.85564 | 0.85135 | 0.84698 | 0.85716 | 0.85383 | |||
| 76.0 | 0.86168 | 0.85322 | 0.84891 | 0.84455 | 0.85473 | 0.85140 | |||
| 77.0 | 0.85927 | 0.85079 | 0.84647 | 0.84211 | 0.85230 | 0.84895 | |||
| 78.0 | 0.85685 | 0.84835 | 0.84403 | 0.83966 | 0.84985 | 0.84650 | |||
| 79.0 | 0.85422 | 0.84590 | 0.84158 | 0.83720 | 0.84740 | 0.84404 | |||
| 80.0 | 0.85197 | 0.84344 | 0.83911 | 0.83473 | 0.84494 | 0.84157 | |||
| 81.0 | 0.84950 | 0.84096 | 0.83664 | 0.83224 | 0.84245 | 0.83909 | |||
| 82.0 | 0.84702 | 0.83848 | 0.83415 | 0.82974 | 0.83997 | 0.83659 | |||
| 83.0 | 0.84453 | 0.83599 | 0.83164 | 0.82724 | 0.83747 | 0.83408 | |||
| 84.0 | 0.84203 | 0.83348 | 0.82913 | 0.82473 | 0.83496 | 0.83156 | |||
| 85.0 | 0.83951 | 0.83095 | 0.82660 | 0.82220 | 0.83242 | 0.82902 | |||
| 86.0 | 0.83697 | 0.82840 | 0.82405 | 0.81965 | 0.82987 | 0.82646 | |||
| 87.0 | 0.83441 | 0.82323 | 0.82148 | 0.81708 | 0.82729 | 0.82389 | |||
| 88.0 | 0.83181 | 0.82323 | 0.81888 | 0.81448 | 0.82469 | 0.82128 | |||
| 89.0 | 0.82919 | 0.82062 | 0.81626 | 0.81186 | 0.82207 | 0.81865 | |||
| 90.0 | 0.82654 | 0.81797 | 0.81362 | 0.80922 | 0.81942 | 0.81600 | |||
| 91.00 | 94.00 | 74.62 | 0.82386 | 0.81529 | 0.81094 | 0.80655 | 0.81674 | 0.81331 | |
| 92.0 | 0.82114 | 0.81257 | 0.80823 | 0.80384 | 0.81401 | 0.81060 | |||
| 93.0 | 0.81839 | 0.80983 | 0.80549 | 0.80111 | 0.81127 | 0.80785 | |||
| 94.0 | 0.81561 | 0.80705 | 0.80272 | 0.79835 | 0.80848 | 0.80507 | |||
| 95.0 | 0.81278 | 0.80424 | 0.79991 | 0.79555 | 0.80567 | 0.80225 | |||
| 96.0 | 0.80991 | 0.80138 | 0.79706 | 0.79271 | 0.80280 | 0.79939 | |||
| 97.0 | 0.80698 | 0.79846 | 0.79415 | 0.78981 | 0.79988 | 0.79648 | |||
| 98.0 | 0.80399 | 0.79547 | 0.79117 | 0.78684 | 0.79688 | 0.79349 | |||
| 99.0 | 0.80094 | 0.79243 | 0.78814 | 0.78382 | 0.79383 | 0.79045 | |||
| 100.0 | 100.0 | 79.39 | 0.79784 | 0.78934 | 0.78506 | 0.78075 | 0.79074 | 0.78736 | −114.3 °C |
| % wt ethanol | % vol ethanol | grams ethanol per 100 cc 15.56°C | d 10°C/4°C | d 20°C/4°C | d 25°C/4°C | d 30°C/4°C | d 20°C/20°C | d 25°C/25°C | freezing temp. |
Properties of aqueous methanol solutions
| % wt methanol | % vol methanol | d 15.6°C/4°C | d 0°C/4°C | d 10°C/4°C | d 20°C/4°C | freezing temp °C |
| 0 | 0 | 0.99908 | 0.99984 | 0.99970 | 0.99820 | 0.0 |
| 1 | 1.25 | 0.99728 | 0.9981 | 0.9980 | 0.9965 | |
| 2 | 2.50 | 0.99543 | 0.9963 | 0.9962 | 0.9943 | |
| 3 | 3.75 | 0.99370 | 0.9946 | 0.9945 | 0.9931 | |
| 3.9 | 5 | 0.9938 | –2.2 | |||
| 4 | 4.99 | 0.99198 | 0.9930 | 0.9929 | 0.9914 | |
| 5 | 6.22 | 0.99029 | 0.9914 | 0.9912 | 0.9896 | |
| 6 | 7.45 | 0.98864 | 0.9899 | 0.9896 | 0.9880 | |
| 7 | 8.68 | 0.98701 | 0.9884 | 0.9881 | 0.9863 | |
| 8 | 9.91 | 0.98547 | 0.9870 | 0.9865 | 0.9847 | |
| 8.1 | 10 | 0.9872 | –5.0 | |||
| 9 | 11.13 | 0.98547 | 0.9856 | 0.9849 | 0.9831 | |
| 10 | 12.35 | 0.98241 | 0.9842 | 0.9834 | 0.9815 | |
| 11 | 13.56 | 0.98093 | 0.9829 | 0.9820 | 0.9799 | |
| 12 | 14.77 | 0.97945 | 0.9816 | 0.9805 | 0.9784 | |
| 12.2 | 15 | 0.9810 | –8.3 | |||
| 13 | 15.98 | 0.97802 | 0.9804 | 0.9791 | 0.9768 | |
| 14 | 17.18 | 0.97660 | 0.9792 | 0.9778 | 0.9754 | |
| 15 | 18.38 | 0.97518 | 0.9780 | 0.9764 | 0.9740 | |
| 16 | 19.58 | 0.97377 | 0.9769 | 0.9751 | 0.9725 | |
| 16.4 | 20 | 0.975 | –11.7 | |||
| 17 | 20.77 | 0.97237 | 0.9758 | 0.9739 | 0.9710 | |
| 18 | 21.96 | 0.97096 | 0.9747 | 0.9726 | 0.9696 | |
| 19 | 23.15 | 0.96955 | 0.9736 | 0.9713 | 0.9681 | |
| 20 | 24.33 | 0.96814 | 0.9725 | 0.9700 | 0.9666 | |
| 20.6 | 25 | 0.968 | –15.6 | |||
| 21 | 25.51 | 0.96673 | 0.9714 | 0.9687 | 0.9651 | |
| 22 | 26.69 | 0.96533 | 0.9702 | 0.9673 | 0.9636 | |
| 23 | 27.86 | 0.96392 | 0.9690 | 0.9660 | 0.9622 | |
| 24 | 29.03 | 0.96251 | 0.9678 | 0.9646 | 0.9607 | |
| 24.9 | 30 | 0.964 | –20.0 | |||
| 25 | 30.19 | 0.96108 | 0.9666 | 0.9632 | 0.9592 | |
| 26 | 31.35 | 0.95963 | 0.9654 | 0.9628 | 0.9576 | |
| 27 | 32.51 | 0.95817 | 0.9642 | 0.9604 | 0.9562 | |
| 28 | 33.66 | 0.95668 | 0.9629 | 0.9590 | 0.9546 | |
| 29 | 34.81 | 0.95518 | 0.9616 | 0.9575 | 0.9531 | |
| 29.2 | 35 | 0.957 | –25.0 | |||
| 30 | 35.95 | 0.95366 | 0.9604 | 0.9560 | 0.9515 | |
| 31 | 37.09 | 0.95213 | 0.9590 | 0.9546 | 0.9499 | |
| 32 | 38.22 | 0.95056 | 0.9576 | 0.9531 | 0.9483 | |
| 33 | 39.35 | 0.94896 | 0.9563 | 0.9516 | 0.9466 | |
| 33.6 | 40 | 0.950 | –30.0 | |||
| 34 | 40.48 | 0.94734 | 0.9549 | 0.9500 | 0.9450 | |
| 35 | 41.59 | 0.94570 | 0.9534 | 0.9484 | 0.9433 | |
| 36 | 42.71 | 0.94404 | 0.9520 | 0.9469 | 0.9416 | |
| 37 | 43.82 | 0.94237 | 0.9505 | 0.9453 | 0.9398 | |
| 38 | 44.92 | 0.94067 | 0.9490 | 0.9437 | 0.9381 | –35.6 |
| 39 | 46.02 | 0.93894 | 0.9475 | 0.9420 | 0.9363 | |
| 40 | 47.11 | 0.93720 | 0.9459 | 0.9403 | 0.9345 | |
| 41 | 48.20 | 0.93543 | 0.9443 | 0.9387 | 0.9327 | |
| 42 | 49.28 | 0.93365 | 0.9427 | 0.9370 | 0.9309 | |
| 43 | 50.35 | 0.93185 | 0.9411 | 0.9352 | 0.9290 | |
| 44 | 51.42 | 0.93001 | 0.9395 | 0.9334 | 0.9272 | |
| 45 | 52.49 | 0.92815 | 0.9377 | 0.9316 | 0.9252 | |
| 46 | 53.54 | 0.92627 | 0.9360 | 0.9298 | 0.9234 | |
| 47 | 54.60 | 0.92436 | 0.9342 | 0.9279 | 0.9214 | |
| 48 | 55.64 | 0.92242 | 0.9324 | 0.9260 | 0.9196 | |
| 49 | 56.68 | 0.92048 | 0.9306 | 0.9240 | 0.9176 | |
| 50 | 57.71 | 0.91852 | 0.9287 | 0.9221 | 0.9156 | |
| 51 | 58.74 | 0.91653 | 0.9269 | 0.9202 | 0.9135 | |
| 52 | 59.76 | 0.91451 | 0.9250 | 0.9182 | 0.9114 | |
| 53 | 60.77 | 0.91248 | 0.9230 | 0.9162 | 0.9094 | |
| 54 | 61.78 | 0.91044 | 0.9211 | 0.9142 | 0.9073 | |
| 55 | 62.78 | 0.90839 | 0.9191 | 0.9122 | 0.9052 | |
| 56 | 63.78 | 0.90631 | 0.9172 | 0.9101 | 0.9032 | |
| 57 | 64.77 | 0.90421 | 0.9151 | 0.9080 | 0.9010 | |
| 58 | 65.75 | 0.90210 | 0.9131 | 0.9060 | 0.8988 | |
| 59 | 66.73 | 0.89996 | 0.9111 | 0.9039 | 0.8968 | |
| 60 | 67.69 | 0.89781 | 0.9090 | 0.9018 | 0.8946 | |
| 61 | 68.65 | 0.89563 | 0.9068 | 0.8998 | 0.8924 | |
| 62 | 69.61 | 0.89341 | 0.9046 | 0.8977 | 0.8902 | |
| 63 | 70.55 | 0.89117 | 0.9024 | 0.8955 | 0.8879 | |
| 64 | 71.49 | 0.88890 | 0.9002 | 0.8933 | 0.8856 | |
| 65 | 72.42 | 0.88662 | 0.8980 | 0.8911 | 0.8834 | |
| 66 | 73.34 | 0.88433 | 0.8958 | 0.8888 | 0.8811 | |
| 67 | 74.26 | 0.88203 | 0.8935 | 0.8865 | 0.8787 | |
| 68 | 75.17 | 0.87971 | 0.8913 | 0.8842 | 0.8763 | |
| 69 | 76.08 | 0.87739 | 0.8891 | 0.8818 | 0.8738 | |
| 70 | 76.98 | 0.87507 | 0.8869 | 0.8794 | 0.8715 | |
| 71 | 77.86 | 0.87271 | 0.8847 | 0.8770 | 0.8690 | |
| 72 | 78.75 | 0.87033 | 0.8824 | 0.8747 | 0.8665 | |
| 73 | 79.62 | 0.86792 | 0.8801 | 0.8724 | 0.8641 | |
| 74 | 80.48 | 0.86546 | 0.8778 | 0.8699 | 0.8616 | |
| 75 | 81.34 | 0.86300 | 0.8754 | 0.8676 | 0.8592 | |
| 76 | 82.18 | 0.86051 | 0.8729 | 0.8651 | 0.8567 | |
| 77 | 83.02 | 0.85801 | 0.8705 | 0.8626 | 0.8542 | |
| 78 | 83.86 | 0.85551 | 0.8680 | 0.8602 | 0.8518 | |
| 79 | 84.68 | 0.85300 | 0.8657 | 0.8577 | 0.8494 | |
| 80 | 85.50 | 0.85048 | 0.8634 | 0.8551 | 0.8469 | |
| 81 | 86.31 | 0.84794 | 0.8610 | 0.8527 | 0.8446 | |
| 82 | 87.11 | 0.84536 | 0.8585 | 0.8501 | 0.8420 | |
| 83 | 87.90 | 0.84274 | 0.8560 | 0.8475 | 0.8394 | |
| 84 | 88.68 | 0.84009 | 0.8535 | 0.8449 | 0.8366 | |
| 85 | 89.45 | 0.83742 | 0.8510 | 0.8422 | 0.8340 | |
| 86 | 90.21 | 0.83475 | 0.8483 | 0.8394 | 0.8314 | |
| 87 | 90.97 | 0.83207 | 0.8456 | 0.8367 | 0.8286 | |
| 88 | 91.72 | 0.82937 | 0.8428 | 0.8340 | 0.8258 | |
| 89 | 92.46 | 0.82667 | 0.8400 | 0.8314 | 0.8230 | |
| 90 | 93.19 | 0.82396 | 0.8374 | 0.8287 | 0.8202 | |
| 91 | 93.92 | 0.82124 | 0.8347 | 0.8261 | 0.8174 | |
| 92 | 94.63 | 0.81849 | 0.8320 | 0.8234 | 0.8146 | |
| 93 | 95.33 | 0.81568 | 0.8293 | 0.8208 | 0.8118 | |
| 94 | 96.02 | 0.81285 | 0.8266 | 0.8180 | 0.8090 | |
| 95 | 96.70 | 0.80999 | 0.8240 | 0.8152 | 0.8062 | |
| 96 | 97.37 | 0.80713 | 0.8212 | 0.8124 | 0.8034 | |
| 97 | 98.04 | 0.80428 | 0.8186 | 0.8096 | 0.8005 | |
| 98 | 98.70 | 0.80143 | 0.8158 | 0.8068 | 0.7976 | |
| 99 | 99.35 | 0.79859 | 0.8130 | 0.8040 | 0.7948 | |
| 100 | 100 | 0.79577 | 0.8102 | 0.8009 | 0.7917 | –97.8 |
| % wt methanol | % vol methanol | d 15.6°C/4°C | d 0°C/4°C | d 10°C/4°C | d 20°C/4°C | freezing temp °C |
Carbon dioxide tables
Solubility in water at various temperatures
| Aqueous Solubility of CO2 at 760 mm Hg pressure | ||
| Temperature | Dissolved CO2 volume at 0 °C, 760 mm Hg per volume H2O | grams/100 ml H2O |
| 0 °C | 1.713 | 0.3346 |
| 1 °C | 1.646 | 0.3213 |
| 2 °C | 1.527 | 0.3091 |
| 3 °C | 1.527 | 0.2978 |
| 4 °C | 1.473 | 0.2871 |
| 5 °C | 1.424 | 0.2774 |
| 6 °C | 1.377 | 0.2681 |
| 7 °C | 1.331 | 0.2589 |
| 8 °C | 1.282 | 0.2492 |
| 9 °C | 1.237 | 0.2403 |
| 10 °C | 1.194 | 0.2318 |
| 11 °C | 1.154 | 0.2239 |
| 12 °C | 1.117 | 0.2165 |
| 13 °C | 1.083 | 0.2098 |
| 14 °C | 1.050 | 0.2032 |
| 15 °C | 1.019 | 0.1970 |
| 16 °C | 0.985 | 0.1903 |
| 17 °C | 0.956 | 0.1845 |
| 18 °C | 0.928 | 0.1789 |
| 19 °C | 0.902 | 0.1737 |
| 20 °C | 0.878 | 0.1688 |
| 21 °C | 0.854 | 0.1640 |
| 22 °C | 0.829 | 0.1590 |
| 23 °C | 0.804 | 0.1540 |
| 24 °C | 0.781 | 0.1493 |
| 25 °C | 0.759 | 0.1449 |
| 26 °C | 0.738 | 0.1406 |
| 27 °C | 0.718 | 0.1366 |
| 28 °C | 0.699 | 0.1327 |
| 29 °C | 0.682 | 0.1292 |
| 30 °C | 0.655 | 0.1257 |
| 35 °C | 0.592 | 0.1105 |
| 40 °C | 0.530 | 0.0973 |
| 45 °C | 0.479 | 0.0860 |
| 50 °C | 0.436 | 0.0761 |
| 60 °C | 0.359 | 0.0576 |
![{\displaystyle \scriptstyle {\frac {[C_{2}H_{5}O^{-}][H^{+}]}{[C_{2}H_{5}OH]}}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/4a1a83514d3f057bba95f26abcd385efd5b98a91)
![{\displaystyle \scriptstyle [H^{+}][OH^{-}]=1.0\times 10^{-14}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/a99b5f9b671eb27da64728d965b1c98ac4653a85)
![{\displaystyle \scriptstyle K_{sp}=[Ca^{2+}][CO_{3}^{2-}]=4.47\times 10^{-9}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/3a790db3573058c9fb0cdcf8ccccf4c0d85ec16f)
![{\displaystyle \scriptstyle {\frac {[CO_{2}]}{p_{CO_{2}}}}={\frac {1}{k'_{c}}}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/e88c3aa916bab6f3dbdeb9d0ecf4c2d2749b8107)

![{\displaystyle \scriptstyle K_{h}={\frac {[H_{2}CO_{3}]}{[CO_{2}]}}=1.70\times 10^{-3}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/c7b96e4cb57576eb8501857b5758cd5ac3407560)
![{\displaystyle \scriptstyle K_{a1}={\frac {[H^{+}][HCO_{3}^{-}]}{[H_{2}CO_{3}]}}=2.5\times 10^{-4}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/43d8e185641bf5361387aa58d754858e826ae944)
![{\displaystyle \scriptstyle K_{a2}={\frac {[H^{+}][CO_{3}^{2-}]}{[HCO_{3}^{-}]}}=5.61\times 10^{-11}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/dc3d142d0ec719145e7cc7bae96c8d6f89fd83f2)
![{\displaystyle \scriptstyle [H^{+}][OH^{-}]=10^{-14}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/825549f914af9988f2472e6fc0b353731714d0aa)
![{\displaystyle \scriptstyle 2[Ca^{2+}]+[H^{+}]=[HCO_{3}^{-}]+2[CO_{3}^{2-}]+[OH^{-}]}](http://wikimedia.org/api/rest_v1/media/math/render/svg/57a73dfcfc3467c382e9d15a4c691e2525e16ef5)


![{\displaystyle \scriptstyle [H^{+}]\simeq \left({\frac {K_{a1}^{2}K_{a2}K_{h}^{2}}{2K_{sp}k_{c}^{\prime 2}}}\right)^{1/3}p_{CO_{2}}^{2/3}\;\;\;,\;\;\;[Ca^{2+}]\simeq \left({\frac {K_{a1}K_{sp}K_{h}}{4K_{a2}k_{c}^{\prime }}}\right)^{1/3}p_{CO_{2}}^{1/3}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/48f647f1b6040a7d4948759af79cf663fbc2dca3)
![{\displaystyle {\frac {P_{\mathrm {CO} _{2}}}{[\mathrm {CO} _{2}]}}\ =\ k'_{c}}](http://wikimedia.org/api/rest_v1/media/math/render/svg/a504a37ec8f4a046866e3c8b81d32de25c786276)









 ,
,  ...
...