User:Snoteleks/Protista
From Wikipedia, the free encyclopedia
Short version
More information Subdomain, Supergroup ...
| Subdomain | Supergroup | Example | Brief description of morphology, lifestyle and habitat | Living species |
|---|---|---|---|---|
| Diaphoretickes | Stramenopiles |  |
Ancestrally flagellates distinguished by two 'heterokont' (unequal) flagella, one with tripartite mastigonemes. The most species-rich lineage, the ochrophytes, are algae of diverse morphologies, ranging from flagellates (like golden algae) to walled ornamented cells (like diatoms, pictured) to truly multicellular macroalgae with differentiated tissues (brown algae such as kelp). All other lineages are composed of heterotrophs: bacterivorous flagellates (e.g., bicosoecids, bigyromonads), fungus-like osmotrophs (oomycetes, hyphochytrids, and labyrinthulomycetes), heliozoan amoebae (actinophryids), and ciliate-like obligate symbionts of animals (opalinids).[1] | over 100,000[2] |
| Alveolata |  |
Ancestrally flagellated predators with cortical alveoli. The colponemids represent these ancestral characteristics.[3] The most diverse group are the ciliates (pictured), with large cells covered in rows of cilia, usually at the top of the microbial food chain.[4] The remaining alveolates belong to the clade Myzozoa and are ancestrally photosynthetic; some have retained their photosynthetic ability (chromerids and many dinoflagellates), while others have evolved into parasites of animals and algae (apicomplexans, perkinsozoans, and some dinoflagellates).[5][6][7] | over 10,000[3] | |
| Rhizaria |  |
Amoebae with fine pseudopodia. The most species-rich group is Retaria, home to conspicuous marine amoebae encased in hard skeletons (radiolarians) or multichambered tests (foraminifers, pictured). Secondly is Cercozoa, especially abundant in soils and exhibiting a wide range of morphologies from amoeboflagellates to aggregative slime molds, testate amoebae (e.g., euglyphids), and radiolarian-like organisms (phaeodarians); some are capable of photosynthesis (e.g., chlorarachniophytes). Lastly, Endomyxa contains both free-living predatory amoebae (e.g., vampyrellids) and obligate parasites of animals, plants, and algae (e.g., phytomyxeans and ascetosporeans).[8][5] | over 11,000[9] | |
| Telonemia |  |
Free-living flagellates with a unique cytoskeleton and a combination of cell structures. Present in all marine and freshwater environments feeding on bacteria.[10] | 10[11][12] | |
| Haptista |  |
Two groups of different free-living single-celled protists: centrohelids—predatory heliozoan amoebae, widespread in aquatic and soil environments[13]—and haptophytes—coccoid or flagellated photosynthetic algae, mostly marine (e.g., coccolithophores, pictured).[14] Both can produce an outer coat of complex mineralized scales.[5] | over 600 | |
| Pancryptista |  |
Free-living flagellates, except one species of heliozoan amoebae, Microheliella maris.[15] Almost all of the flagellates are distinguished by specialized ribbon-shaped extrusomes known as ejectisomes. Many are photosynthetic, known as cryptomonads (pictured), while the rest are phagotrophs, consumers of bacteria. Present in aquatic environments worldwide.[16][5] | over 100 | |
| Archaeplastida* |  |
Algae with chloroplasts derived from primary endosymbiosis with a cyanobacterium. Found in all environments. Almost entirely photosynthetic, with the exception of two small groups of phagotrophic flagellates, rhodelphids and picozoans. The two major groups, red algae and green algae (pictured), exhibit diverse morphologies, ranging from single cells—coccoid, palmelloid, sarcinoid, flagellated—to colonies, simple filaments, and macroscopic thalli with varying degrees of complexity (e.g., coralline algae, sea lettuce, stoneworts). Also included are glaucophytes, rare blue-green algae found in surface waters.[17] | over 20,500[18]* | |
| Disparia |  |
Three lineages of free-living predatory flagellates with unique cytoskeletons. These are: Hemimastigophora, with two rows of flagella, present in soils and aquatic sediments; Provora, fast-swimming predators of other protists through a strong feeding apparatus resembling jaws, found in low abundance in marine environments globally;[19] and Caelestes (pictured), rare inhabitants of the marine benthos whose cells protrude arms or stalks used for movement or prey capture.[20] | 20 | |
| Amorphea | Amoebozoa |  |
Amoebae of diverse morphologies, with blunt (lobose) or fine (filose) pseudopodia, and sometimes with flagella. Most are free-living phagotrophs found across terrestrial and aquatic environments, such as the archetypal genus Amoeba itself,[21] or the testate amoebae Arcellinida, one of the most conspicuous groups of protists.[22] Several groups have independently evolved the ability to form fungus-like fruiting bodies, such as myxomycetes (pictured).[23] Some of the free-living amoebae are important vectors of pathogenic bacteria or are pathogenic themselves (e.g., Acanthamoeba).[24] Others are anaerobic intestinal symbionts (e.g., Entamoeba).[5] | over 2,400[25] |
| Breviatea |  |
Anaerobic free-living amoeboflagellates with fine pseudopodia and modified mitochondria. Present only in low-oxygen marine and brackish sediments, their growth depends on mutualistic interactions with prokaryotes.[26] | 4[27] | |
| Apusomonadida |  |
Free-living flagellates distinguished by a proboscis, a sleeve-like structure that envelops one of their two flagella.[27] Found gliding on wet soil and aquatic sediments worldwide.[28] | 28[28] | |
| Opisthokonta** |  |
Flagellates distinguished by a single posterior flagellum, many with complex life cycles and varying degrees of multicellularity.[23] Some are entirely amoeboid, with fine pseudopodia (e.g., filastereans and nucleariids, including slime molds),[29][30] while others become amoeboid temporarily (e.g., choanoflagellates, pictured).[31] Most species are free-living filter-feeders or predators,[32][30][33][34] but some lineages (e.g., ichthyosporids) evolved into osmotrophic parasites of animals.[35][36] | approx. 300[9]** | |
| Excavates | Discoba |  |
Flagellates with very different lifestyles, present in aquatic and terrestrial environments, ranging from aerobes to anaerobes. The most diverse group, Euglenozoa, includes free-living osmotrophs, phagotrophs, phototrophs (euglenophytes, pictured), and pathogens (kinetoplastids).[37] The less diverse Heterolobosea are primarily amoeboflagellates, and include some slime molds (acrasids) and well-known opportunistic parasites (e.g., Naegleria fowleri).[38] The smallest group, Jakobida, consume bacteria by suspension feeding.[39] | over 2,200[40][41] |
| Metamonada |  |
Anaerobic or microaerophilic flagellates, amoebae, or amoeboflagellates,[42] with reduced or completely lost[43] mitochondria. A few are free-living, found in aquatic hypoxic sediments, but most species are obligate parasites (e.g., Giardia, pictured) or commensals in animal intestines (e.g., parabasalids). Many have a high number of flagella.[44][42][5] | approx. 800[9] | |
| Malawimonadida | 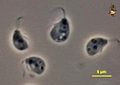 |
Free-living bacterivorous flagellates that feed by suspension feeding, present in marine or fresh waters.[45] | 3[45] | |
| Other | Ancyromonadida | Tiny free-living aquatic flagellates composed of flattened cells with an inflexible pellicle and a lateral rostrum with extrusomes. Found in most aquatic habitats.[46] | over 20[27][46] | |
| CRuMs |  |
Free-living flagellates and filose amoebae with a pellicle underneath the cell membrane. Almost all flagellated members can produce filose pseudopodia. Found in aquatic environments.[47] | 14[47] | |
| *Excluding plants. **Excluding animals and fungi. | ||||
Close
- Jirsová, Dagmar; Wideman, Jeremy G (8 January 2024). "Integrated overview of stramenopile ecology, taxonomy, and heterotrophic origin". The ISME Journal. 18 (1) wrae150. doi:10.1093/ismejo/wrae150. ISSN 1751-7362. PMC 11412368. PMID 39077993.
- H.S. Yoon; R.A. Andersen; S.M. Boo; D. Bhattacharya (17 February 2009). "Stramenopiles". Encyclopedia of Microbiology (Third ed.). pp. 721–731. doi:10.1016/B978-012373944-5.00253-4. ISBN 978-0-12-373944-5. Retrieved 2 March 2024.
- Tikhonenkov, Denis V.; Strassert, Jürgen F.H.; Janouškovec, Jan; Mylnikov, Alexander P.; Aleoshin, Vladimir V.; Burki, Fabien; Keeling, Patrick J. (2020). "Predatory colponemids are the sister group to all other alveolates". Molecular Phylogenetics and Evolution. 149: 106839. doi:10.1016/j.ympev.2020.106839. Retrieved 19 February 2026.
{{cite journal}}: CS1 maint: article number as page number (link) - H. Lynn, Denis (2017). "Ciliophora". In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists (PDF). Vol. 1 (2nd ed.). Cham: Springer International Publishing. pp. 679–730. doi:10.1007/978-3-319-28149-0_23. ISBN 978-3-319-28149-0. LCCN 2017945328.
- Mathur, Varsha; Kolísko, Martin; Hehenberger, Elisabeth; Irwin, Nicholas A.T.; Leander, Brian S.; et al. (2019). "Multiple Independent Origins of Apicomplexan-Like Parasites". Current Biology. 29 (17): 2936–2941. doi:10.1016/j.cub.2019.07.019. PMID 31422883.
- Holt, Corey C.; Hehenberger, Elisabeth; Tikhonenkov, Denis V.; Jacko-Reynolds, Victoria K. L.; Okamoto, Noriko; Cooney, Elizabeth C.; Irwin, Nicholas A. T.; Keeling, Patrick J. (3 November 2023). "Multiple parallel origins of parasitic Marine Alveolates". Nature Communications. 14 (1) 7049. doi:10.1038/s41467-023-42807-0. ISSN 2041-1723. PMC 10624901. PMID 37923716.
- Burki, Fabien; Keeling, Patrick J. (2014). "Rhizaria". Current Biology. 24 (3): R103–R107. doi:10.1016/j.cub.2013.12.025.
- Adl, Sina M.; Leander, Brian S.; Simpson, Alastair G. B.; Archibald, John M.; Anderson, O. Roger.; et al. (1 August 2007). "Diversity, Nomenclature, and Taxonomy of Protists" (PDF). Systematic Biology. 56 (4): 684–689. doi:10.1080/10635150701494127. ISSN 1076-836X. PMID 17661235. Retrieved 15 July 2025.
- Tikhonenkov, Denis V.; Jamy, Mahwash; Borodina, Anastasia S.; Belyaev, Artem O.; Zagumyonnyi, Dmitry G.; Prokina, Kristina I.; Mylnikov, Alexander P.; Burki, Fabien; Karpov, Sergey A. (2022). "On the origin of TSAR: morphology, diversity and phylogeny of Telonemia". Open Biology. 12 (3). The Royal Society. doi:10.1098/rsob.210325. ISSN 2046-2441. PMC 8924772. PMID 35291881.
- Zlatogursky, Vasily; Boscaro, Vittorio; Lax, Gordon; Wanntorp, Matias; Pohl, Nina; Burki, Fabien; Keeling, Patrick J. (15 August 2025). "Phylogenetic position and mitochondrial genome evolution of "orphan" eukaryotic lineages". iScience. 28 (8) 113184. doi:10.1016/j.isci.2025.113184. PMC 12432456. PMID 40948565.
- Mostazo‐Zapata, Helena; Gàlvez‐Morante, Alex; Berney, Cédric; Maya‐Figuerola, Xènia; Sigona, Cristiana; López‐Escardó, David; Sà, Elisabet L.; Vaqué, Dolors; Richter, Daniel J. (2025). "Description of a New Telonemia Genus and Species With Novel Observations Providing Insights Into Its Hidden Diversity". Journal of Eukaryotic Microbiology. 72 (6) e70050. doi:10.1111/jeu.70050. ISSN 1066-5234. PMC 12550356. PMID 41131848.
- Zagumyonnyi, Dmitry G.; Radaykina, Liudmila V.; Tikhonenkov, Denis V. (11 December 2021). "Triangulopteris lacunata gen. et sp. nov. (Centroplasthelida), a New Centrohelid Heliozoan from Soil". Diversity. 13 (12) 658. doi:10.3390/d13120658. ISSN 1424-2818.
- Eikrem, Wenche; Medlin, Linda K.; Henderiks, Jorijntje; Rokitta, Sebastian; Rost, Björn; et al. (2017). "Haptophyta" (PDF). In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists (PDF). Vol. 2 (2nd ed.). Cham: Springer International Publishing. pp. 893–953. doi:10.1007/978-3-319-28149-0_38. ISBN 978-3-319-28149-0. LCCN 2017945328. Retrieved 9 June 2025.
- Yazaki, Euki; Yabuki, Akinori; Imaizumi, Ayaka; Kume, Keitaro; Hashimoto, Tetsuo; Inagaki, Yuji (2022-04-13). "The closest lineage of Archaeplastida is revealed by phylogenomics analyses that include Microheliella maris". Open Biology. 12 (4) 210376. doi:10.1098/rsob.210376. PMC 9006020. PMID 35414259.
- Hoef-Emden, Kerstin; Archibald, John M. (2017). "Cryptophyta (Cryptomonads)". In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists (PDF). Vol. 2 (2nd ed.). Cham: Springer International Publishing. pp. 851–891. doi:10.1007/978-3-319-28149-0_35. ISBN 978-3-319-28149-0. LCCN 2017945328.
- Price, Dana C.; Steiner, Jürgen M.; Yoon, Hwan Su; Bhattacharya, Debashish; Löffelhardt, Wolfgang (2017). "Glaucophyta" (PDF). In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists (PDF). Vol. 1 (2nd ed.). Cham: Springer International Publishing. pp. 23–87. doi:10.1007/978-3-319-28149-0_42. ISBN 978-3-319-28149-0. LCCN 2017945328. Retrieved 9 June 2025.
- Bowles, Alexander M.C.; Williamson, Christopher J.; Williams, Tom A.; Lenton, Timothy M.; Donoghue, Philip C.J. (2023). "The origin and early evolution of plants". Trends in Plant Science. 28 (3): 312–329. doi:10.1016/j.tplants.2022.09.009.
- Tikhonenkov, Denis V.; Mikhailov, Kirill V.; Gawryluk, Ryan M. R.; Belyaev, Artem O.; Mathur, Varsha; et al. (22 December 2022). "Microbial predators form a new supergroup of eukaryotes". Nature. 612 (7941): 714–719. doi:10.1038/s41586-022-05511-5. PMID 36477531.
- Valt, Marek; Pánek, Tomáš; Mirzoyan, Seda; Tice, Alexander K.; Jones, Robert E.; et al. (19 November 2025). "Rare microbial relict sheds light on an ancient eukaryotic supergroup" (PDF). Nature. doi:10.1038/s41586-025-09750-0. ISSN 0028-0836. Retrieved 25 November 2025.
- Kang, Seungho; Tice, Alexander K; Spiegel, Frederick W; Silberman, Jeffrey D; Pánek, Tomáš; et al. (September 2017). "Between a Pod and a Hard Test: The Deep Evolution of Amoebae". Molecular Biology and Evolution. 34 (9): 2258–2270. doi:10.1093/molbev/msx162. PMC 5850466. PMID 28505375.
- González-Miguéns, Rubén; Todorov, Milcho; Blandenier, Quentin; Duckert, Clément; Porfirio-Sousa, Alfredo L.; Ribeiro, Giulia M.; Ramos, Diana; Lahr, Daniel J.G.; Buckley, David; Lara, Enrique (2022). "Deconstructing Difflugia: The tangled evolution of lobose testate amoebae shells (Amoebozoa: Arcellinida) illustrates the importance of convergent evolution in protist phylogeny". Molecular Phylogenetics and Evolution. 175 107557. Bibcode:2022MolPE.17507557G. doi:10.1016/j.ympev.2022.107557. hdl:10261/281619. PMID 35777650.
- Lamża, Łukasz (2023). "Diversity of 'simple' multicellular eukaryotes: 45 independent cases and six types of multicellularity". Biological Reviews. 98 (6): 2188–2209. doi:10.1111/brv.13001. ISSN 1464-7931.
- Martinez, Augusto Julio; Visvesvara, Govinda S. (1997). "Free‐living, Amphizoic and Opportunistic Amebas". Brain Pathology. 7 (1): 583–598. doi:10.1111/j.1750-3639.1997.tb01076.x. ISSN 1015-6305. PMC 8098488. PMID 9034567.
- Pawlowski, Jan; Audic, Stéphane; Adl, Sina; Bass, David; Belbahri, Lassaâd; et al. (6 November 2012). "CBOL Protist Working Group: Barcoding Eukaryotic Richness beyond the Animal, Plant, and Fungal Kingdoms". PLoS Biology. 10 (11): e1001419. doi:10.1371/journal.pbio.1001419. ISSN 1545-7885. PMC 3491025. PMID 23139639.
{{cite journal}}: CS1 maint: article number as page number (link) - Aguilera-Campos, Karla Iveth; Boisard, Julie; Törnblom, Viktor; Jerlström-Hultqvist, Jon; Behncké-Serra, Ada; Cotillas, Elena Aramendia; Stairs, Courtney Weir (2 January 2025). "Anaerobic breviate protist survival in microcosms depends on microbiome metabolic function". The ISME Journal. 19 (1) wraf171. doi:10.1093/ismejo/wraf171. ISSN 1751-7362. PMC 12453579. PMID 40795332.
- Heiss, Aaron A.; Brown, Matthew W.; Simpson, Alastair G. B. (2017). "Apusomonadida". In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists (PDF). Vol. 2 (2nd ed.). Cham: Springer International Publishing. pp. 1619–1645. doi:10.1007/978-3-319-28149-0_12. ISBN 978-3-319-28149-0. LCCN 2017945328.
- Torruella, Guifré; Galindo, Luis Javier; Moreira, David; Ciobanu, Maria; Heiss, Aaron A.; et al. (2023). "Expanding the molecular and morphological diversity of Apusomonadida, a deep‐branching group of gliding bacterivorous protists". Journal of Eukaryotic Microbiology. 70 (2) e12956. doi:10.1111/jeu.12956.
- Torruella, Guifré; de Mendoza, Alex; Grau-Bové, Xavier; Antó, Meritxell; Chaplin, Mark A.; et al. (2015). "Phylogenomics Reveals Convergent Evolution of Lifestyles in Close Relatives of Animals and Fungi". Current Biology. 25 (18): 2404–2410. doi:10.1016/j.cub.2015.07.053. PMID 26365255.
- Gabaldón, Toni; Völcker, Eckhard; Torruella, Guifré (2022). "On the Biology, Diversity and Evolution of Nucleariid Amoebae (Amorphea, Obazoa, Opisthokonta)". Protist. 173 (4) 125895. doi:10.1016/j.protis.2022.125895. hdl:2117/369912. PMID 35841659.
- Brunet, Thibaut; Albert, Marvin; Roman, William; Coyle, Maxwell C; Spitzer, Danielle C; King, Nicole (15 January 2021). "A flagellate-to-amoeboid switch in the closest living relatives of animals". eLife. 10 e61037. doi:10.7554/eLife.61037. ISSN 2050-084X.
- Richter, Daniel J.; Nitsche, Frank (2017). "Choanoflagellatea". In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists. Vol. 2 (2nd ed.). Springer. pp. 1479–1496. doi:10.1007/978-3-319-28149-0_5. ISBN 978-3-319-28147-6.
- Hehenberger, Elisabeth; Tikhonenkov, Denis V.; Kolisko, Martin; del Campo, Javier; Esaulov, Anton S.; Mylnikov, Alexander P.; Keeling, Patrick J. (10 July 2017). "Novel Predators Reshape Holozoan Phylogeny and Reveal the Presence of a Two-Component Signaling System in the Ancestor of Animals". Current Biology. 27 (13): 2043–2050. Bibcode:2017CBio...27E2043H. doi:10.1016/j.cub.2017.06.006. PMID 28648822.
- Tikhonenkov, Denis V.; Mikhailov, Kirill V.; Hehenberger, Elisabeth; Karpov, Sergei A.; Prokina, Kristina I.; et al. (16 November 2020). "New Lineage of Microbial Predators Adds Complexity to Reconstructing the Evolutionary Origin of Animals". Current Biology. 30 (22): 4500–4509.e5. Bibcode:2020CBio...30E4500T. doi:10.1016/J.CUB.2020.08.061. PMID 32976804.
- Glockling, Sally L.; Marshall, Wyth L.; Gleason, Frank H. (25 April 2013). "Phylogenetic interpretations and ecological potentials of the Mesomycetozoea (Ichthyosporea)". Fungal Ecology. 6 (4): 237–247. doi:10.1016/j.funeco.2013.03.005.
- Urrutia, Ander; Mitsi, Konstantina; Foster, Rachel; Ross, Stuart; Carr, Martin; et al. (2022). "Txikispora philomaios n. sp., n. g., a micro-eukaryotic pathogen of amphipods, reveals parasitism and hidden diversity in Class Filasterea". Journal of Eukaryotic Microbiology. 69 (2) e12875. doi:10.1111/jeu.12875. PMID 34726818.
- Kostygov, Alexei Y.; Karnkowska, Anna; Votýpka, Jan; Tashyreva, Daria; Maciszewski, Kacper; Yurchenko, Vyacheslav; Lukeš, Julius (2021). "Euglenozoa: taxonomy, diversity and ecology, symbioses and viruses". Open Biology. 11 (3) 200407. doi:10.1098/rsob.200407. PMC 8061765. PMID 33715388.
- Pánek, Tomáš; Simpson, Alastair G. B.; Brown, Matthew W.; Dyer, Betsey Dexter (2017). "Heterolobosea". In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists. Vol. 2 (2nd ed.). Springer. pp. 1005–1046. doi:10.1007/978-3-319-28149-0_10. ISBN 978-3-319-28147-6.
- Simpson, Alastair G. B. (2017). "Jakobida". In Archibald, John M.; Simpson, Alastair G.B.; Slamovits, Claudio H. (eds.). Handbook of the Protists. Vol. 2 (2nd ed.). Springer. pp. 973–1004. doi:10.1007/978-3-319-28149-0_6. ISBN 978-3-319-28147-6.
- Guiry, Michael D. (2024). "How many species of algae are there? A reprise. Four kingdoms, 14 phyla, 63 classes and still growing". Journal of Phycology. 60 (2): 214–228. Bibcode:2024JPcgy..60..214G. doi:10.1111/jpy.13431. PMID 38245909.
- Pánek, Tomáš; Zadrobílková, Eliška; Walker, Giselle; Brown, Matthew W.; Gentekaki, Eleni; et al. (2016). "First multigene analysis of Archamoebae (Amoebozoa: Conosa) robustly reveals its phylogeny and shows that Entamoebidae represents a deep lineage of the group". Molecular Phylogenetics and Evolution. 98: 41–51. doi:10.1016/j.ympev.2016.01.011.
- Boscaro, Vittorio; James, Erick R.; Fiorito, Rebecca; del Campo, Javier; Scheffrahn, Rudolf H.; Keeling, Patrick J. (2024). "Updated classification of the phylum Parabasalia". Journal of Eukaryotic Microbiology. 71 (4): e13035. doi:10.1111/jeu.13035. ISSN 1066-5234.
{{cite journal}}: CS1 maint: article number as page number (link) - Karnkowska, Anna; Vacek, Vojtěch; Zubáčová, Zuzana; Treitli, Sebastian C.; Petrželková, Romana; et al. (2016). "A Eukaryote without a Mitochondrial Organelle". Current Biology. 26 (10): 1274–1284. doi:10.1016/j.cub.2016.03.053.
- Yubuki, Naoji; Huang, Sam S.C.; Leander, Brian S. (2016). "Comparative Ultrastructure of Fornicate Excavates, Including a Novel Free-living Relative of Diplomonads: Aduncisulcus paluster gen. et sp. nov". Protist. 167 (6): 584–596. doi:10.1016/j.protis.2016.10.001.
- Heiss, Aaron A.; Warring, Sally D.; Lukacs, Kaleigh; Favate, John; Yang, Ashley; et al. (2021). "Description of Imasa heleensis , gen. nov., sp. nov. (Imasidae, fam. nov.), a Deep‐Branching Marine Malawimonad and Possible Key Taxon in Understanding Early Eukaryotic Evolution". Journal of Eukaryotic Microbiology. 68 (2) e12837. doi:10.1111/jeu.12837.
- Yubuki, Naoji; Torruella, Guifré; Galindo, Luis Javier; Heiss, Aaron A.; Ciobanu, Maria Cristina; et al. (22 August 2023). "Molecular and morphological characterization of four new ancyromonad genera and proposal for an updated taxonomy of the Ancyromonadida". Journal of Eukaryotic Microbiology. 70 (6): e12997. doi:10.1111/jeu.12997. hdl:2117/404022. ISSN 1550-7408. PMID 37606230.
{{cite journal}}: CS1 maint: article number as page number (link) - Yazaki, Euki; Harada, Ryo; Isogai, Ryu; Bamba, Kohei; Ishida, Ken-ichiro; Inagaki, Yuji; Shiratori, Takashi (June 2025). "Glissandra oviformis n. sp.: a novel predatory flagellate illuminates the character evolution within the eukaryotic clade CRuMs". Open Biology. 15 (6) 250057. doi:10.1098/rsob.250057. PMC 12133344. PMID 40460873.
