1,2-Dithietane
Chemical compound
From Wikipedia, the free encyclopedia
1,2-Dithietane is a dithietane. It is a heterocyclic compound with a four-membered ring. Two sulfur atoms are adjacent, and the molecule is saturated. 1,2-Dithietane has not been produced as of 2000. The combination of ring strain, and lone pairs of electrons, which repel each other, on the sulfur atoms makes the sulfur-sulfur bond too weak to produce the molecule. However a few derivatives are known. 3,4-Diethyl-1,2-dithietane 1,1-dioxide has one sulfur fully oxidised. Dithiatopazine is a tricyclic compound with the -S-S- as a bridge.[1][2] 1,2-Dithietan-3-one, the ketone of 1,2-dithietane, was produced in 2008 by reacting α-dithiolactone with ethoxycarbonylformonitrile oxide.[3] 4,4-di-tert-butyl-1,2-dithietan-3-one and the spiro compound 5,5,9,9-tetramethyl-1,2-dithiaspiro[3.5]nonan-3-one have also been made.[3]

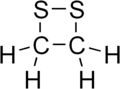 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,2-Dithietane | |
| Systematic IUPAC name
1,2-Dithiacyclobutane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H4S2 | |
| Molar mass | 92.18 g/mol |
| Related compounds | |
Related compounds |
Dithiete, Dioxetane, 1,2-Dithiadistannetane |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
1,2-Dithietane was claimed to have been made by reacting 1,2-ethanedithiol with iodine, but the major product was an eight-membered ring.[4]
A reaction of 2,3-dimercapto-1-propanol with 3-methyllumiflavin transiently produced 3-hydroxy-1,2-dithietane. This dithietane polymerised under light, breaking and reforming the S-S bonds to form a long chain -SSCH(OH)CH2-.[5]
