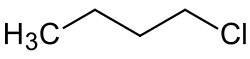
1-Chlorobutane
Chemical compound
From Wikipedia, the free encyclopedia
1-Chlorobutane is an alkyl halide with the chemical formula C
4H
9Cl. It is a colorless, flammable liquid. It is used as a solvent in some industries due to its ability to dissolve both polar and non-polar substances.[4] It is an alkylating reagent, that is used in organic synthesis to add a butyl group to other molecules.[5]

 | |
| Names | |
|---|---|
| Preferred IUPAC name
1-Chlorobutane | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.361 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1127 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H9Cl | |
| Molar mass | 92.57 g·mol−1 |
| Appearance | Colorless liquid[1] |
| Density | 0.89 g/mL |
| Melting point | −123.1 °C (−189.6 °F; 150.1 K)[1] |
| Boiling point | 78 °C (172 °F; 351 K)[1] |
| 0.5 g/L (20 °C)[1] | |
| Solubility | Miscible with methanol, ether[citation needed] |
| log P | 2.56[2] |
| Vapor pressure | 103.4±0.1 mmHg at 25 °C[2] |
| −67.10·10−6 cm3/mol | |
Refractive index (nD) |
1.396[2] |
| Viscosity | 0.4261 mPa·s[3] |
| Hazards | |
| GHS labelling: | |
 | |
| Danger | |
| H225 | |
| P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P370+P378, P403+P235, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | −12 °C (10 °F; 261 K)[1] |
| Safety data sheet (SDS) | Fischer MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Preparation and reactions
It can be prepared from 1-butanol by treatment with hydrogen chloride:[6]
- C4H9OH + HCl → C4H9Cl + H2O
It reacts with lithium metal to give n-butyllithium:[7]
- 2 Li + C
4H
9Cl → C
4H
9Li + LiCl
1-Chlorobutane reacts with sodium hydroxide via nucleophilic substitution to produce butanol:
- C
4H
9Cl + NaOH → C
4H
9OH + NaCl