Acetoacetamide
Chemical compound
From Wikipedia, the free encyclopedia
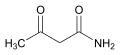
Acetoacetamide is an organic compound with the formula CH3COCH2CONH2. It is the amide of acetoacetic acid. It is produced by treating diketene with aqueous ammonia[1]
 | |
| Names | |
|---|---|
| Other names
3-oxobutanamide, 3-oxobutyramide | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.025.250 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H7NO2 | |
| Molar mass | 101.105 g·mol−1 |
| Appearance | white solid |
| Melting point | 53-56 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
It results from degradation of the sweetner acesulfame potassium.[2]
Related compounds
Acetoacetanilide (CH3COCH2CONH2) is the N-phenyl derivative of acetoacetamide. It is also prepared from diketene.[3] It and various derivatives are used in the production of organic pigments called arylide yellows, one example being Pigment Yellow 74. Many analogues have been prepared.[4] To make the dyes, acetoacetanilides are coupled to diazonium salts, "azo coupling".[5]
