Agaritine
Chemical compound
From Wikipedia, the free encyclopedia
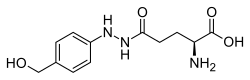
Agaritine is an aromatic hydrazine-derivative mycotoxin in mushroom species of the genus Agaricus.[1] It is an α-aminoacid and a derivative of phenylhydrazine.

 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-[4-(Hydroxymethyl)phenyl]-glutamohydrazide | |
| Systematic IUPAC name
2-Amino-4-{N'-[4-(hydroxymethyl)phenyl]hydrazinecarbonyl}butanoic acid | |
| Other names
β-N-[γ-glutamyl]-4-hydroxymethylphenylhydrazine N2-(γ-glutamyl)-4-hydroxymethylphenylhydrazine | |
| Identifiers | |
3D model (JSmol) |
|
| Abbreviations | AGT |
| 757731 | |
| ChEBI | |
| ChemSpider | |
| KEGG | |
| MeSH | Agaritine |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C12H17N3O4 | |
| Molar mass | 267.285 g·mol−1 |
| Melting point | 203 °C (397 °F; 476 K) |
| Acidity (pKa) | 3.4 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Toxic |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Occurrence
Agaritine is present as a natural mycotoxin in fresh samples of at least 24 species of the genera Agaricus, Leucoagaricus, and Macrolepiota.[2] Mushrooms of these species are found around the world.[3] These mushrooms grow in a wide range of habitats. Agaricus bisporus is cultivated in over 70 countries and on every continent except Antarctica. A. bisporus, also known as the portobello or common button mushroom, is of particular socio-economic importance in developed countries.[4]
Agaritine content varies between individual mushrooms and across species. Agaritine content (% fresh weight) in raw Agaricus bisporus, for example, ranges from 0.033% to 0.173%, with an average of 0.088%. The highest amount of agaritine is found in the cap and gills of the fruiting body, and the lowest in the stem.[2] Agaratine decomposes significantly between harvest and sale, and it has also been shown to decompose readily upon cooking (up to 90% reduction) as well as upon freezing (up to 75% reduction).[5][6][7]
Production
Potential for toxicity
Although recognized as an experimental carcinogen when used in high laboratory doses, there is inadequate evidence to classify Agaritine as carcinogenic to humans in amounts ingested from consuming mushrooms.[1]
