Benzofuran
Heterocyclic compound consisting of fused benzene and furan rings
From Wikipedia, the free encyclopedia
Benzofuran is the heterocyclic compound consisting of fused benzene and furan rings. This colourless liquid is a component of coal tar. Benzofuran is the structural nucleus (parent compound) of many related compounds with more complex structures. For example, psoralen is a benzofuran derivative that occurs in several plants.

 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1-Benzofuran[1] | |||
| Other names | |||
| Identifiers | |||
3D model (JSmol) |
|||
| 107704 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.005.439 | ||
| EC Number |
| ||
| 260881 | |||
| KEGG | |||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1993 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C8H6O | |||
| Molar mass | 118.135 g·molâ1 | ||
| Melting point | â18 °C (0 °F; 255 K) | ||
| Boiling point | 173 °C (343 °F; 446 K) | ||
| Hazards | |||
| GHS labelling: | |||
  | |||
| Warning | |||
| H226, H351, H412 | |||
| P201, P202, P210, P233, P240, P241, P242, P243, P273, P280, P281, P303+P361+P353, P308+P313, P370+P378, P403+P235, P405, P501 | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
500 mg/kg (mice).[2] | ||
| Related compounds | |||
Related compounds |
Benzothiophene, Indole, Indene, 2-Cumaranone | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
There are benzofuran based herbicides: ethofumesate and benfuresate.[3] Several psychoactive drugs such as 5-MAPB and 6-APB are also derived from benzofuran.
Production
Benzofuran is extracted from coal tar. It is also obtained by dehydrogenation of 2-ethylphenol.[2]
Laboratory methods
Benzofurans can be prepared by various methods in the laboratory. Notable examples include:
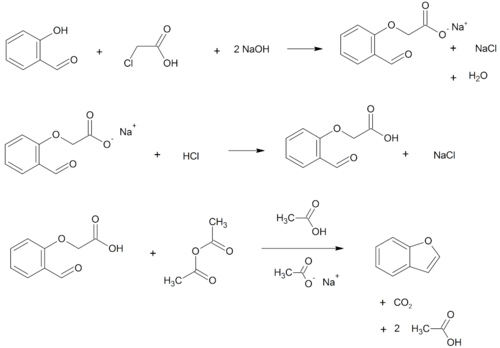
- O-alkylation of salicylaldehyde with chloroacetic acid followed by dehydration (cyclication) of the resulting ether and decarboxylation.[4]
- DielsâAlder reaction[clarification needed] of nitro vinyl furans with various dienophiles:[8]
Related compounds
- Substituted benzofurans
- Dibenzofuran, an analog with a second fused benzene ring.
- Furan, an analog without the fused benzene ring.
- Indole, an analog with a nitrogen instead of the oxygen atom.
- Benzothiophene, an analog with a sulfur instead of the oxygen atom.
- Isobenzofuran, the isomer with oxygen in the adjacent position.
- Aurone
- Thunberginol F