Butalene
From Wikipedia, the free encyclopedia
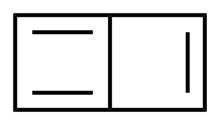 | |
| Names | |
|---|---|
| Preferred IUPAC name
Bicyclo[2.2.0]hexa-1,3,5-triene | |
| Other names
Butalene | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H4 | |
| Molar mass | 76.098 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Butalene is a polycyclic hydrocarbon composed of two fused cyclobutadiene rings.[1] A reported possible synthesis of it involves an elimination reaction from a Dewar benzene derivative. The structure itself can be envisioned as benzene with an internal bridge, and calculations indicate it is somewhat less stable than the open 1,4-didehydrobenzene biradical, the valence isomer in which that bridged bond is broken.
