Formate
Salt or ester of formic acid
From Wikipedia, the free encyclopedia
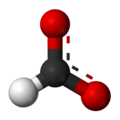
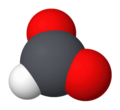
Formate (IUPAC name: methanoate) is the conjugate base of formic acid. Formate is an anion (HCO−2) or its derivatives such as ester of formic acid. The salts and esters are generally colorless.[1]

 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Formate | |||
| Systematic IUPAC name
Methanoate | |||
| Other names
Formylate Methylate Isocarbonite Carbonite(1-) Hydrogencarboxylate Metacarbonoate Oxocarbinate Oxomethyl oxide ion Oxomethoxide | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| 1006 | |||
| KEGG | |||
| MeSH | Formates | ||
PubChem CID |
|||
| UNII | |||
| |||
| |||
| Properties | |||
| HCOO− or HCO− 2 | |||
| Molar mass | 45.017 g mol−1 | ||
| Conjugate acid | Formic acid | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Fundamentals
When dissolved in water, formic acid converts to formate:
- HCO2H → HCO−2 + H+
Formate is a planar anion. The two oxygen atoms are equivalent and bear a partial negative charge. The remaining C-H bond is not acidic.
Biochemistry
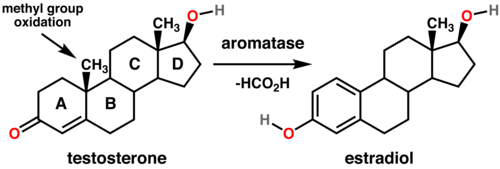
Formate is a common C-1 source in living systems. It is formed from many precursors including choline, serine, and sarcosine. It provides a C-1 source in the biosynthesis of some nucleic acids. Formate (or formic acid) is invoked as a leaving group in the demethylation of some sterols.[2] These conversions are catalyzed by aromatase enzymes using O2 as the oxidant. Specific conversions include testosterone to estradiol and androstenedione to estrone.[3]
Formate is reversibly oxidized by the enzyme formate dehydrogenase from Desulfovibrio gigas:[4]
- HCO−2 → CO2 + H+ + 2 e−
Formate esters
Formate esters have the formula HCOOR (alternative way of writing formula ROC(O)H or RO2CH). Many form spontaneously when alcohols dissolve in formic acid; contrariwise, they hydrolyze easily in base.[5]: 674, 682 Some formate esters arise by the addition of formic acid to alkenes.[6]
An important formate ester is methyl formate, which is produced as an intermediate en route to formic acid. Methanol and carbon monoxide react in the presence of a strong base, such as sodium methoxide:[1]
- CH3OH + CO → HCOOCH3
Hydrolysis of methyl formate gives formic acid and regenerates methanol:
- HCOOCH3 → HCOOH + CH3OH
In laboratory, formate esters can be used to produce pure carbon monoxide.[7]
Reaction with phosphorus pentachloride does not chlorolyze the ester bond, instead forming an (unusual) dichloromethyl ether.[5]: 684
Formate esters often are fragrant or have distinctive odors. Compared to the more common acetate esters, formate esters are less commonly used commercially because they are less stable.[8] Ethyl formate is found in some confectionaries.[1]
Formate salts

Formate salts have the formula M(O2CH)(H2O)x. Such salts are prone to decarboxylation. For example, hydrated nickel formate decarboxylates at about 200 °C with reduction of the Ni2+ to finely powdered nickel metal:
- Ni(HCO2)2(H2O)2 → Ni + 2 CO2 + 2 H2O + H2
Such fine powders are useful as hydrogenation catalysts.[1]
Examples
- ethyl formate, CH3CH2(HCOO)
- sodium formate, Na(HCOO)
- potassium formate, K(HCOO)
- caesium formate, Cs(HCOO); see Caesium: Petroleum exploration
- methyl formate, CH3(HCOO)
- methyl chloroformate, CH3OCOCl
- triethyl orthoformate
- trimethyl orthoformate, C4H10O3
- phenyl formate HCOOC6H5
- amyl formate