Coble creep
From Wikipedia, the free encyclopedia
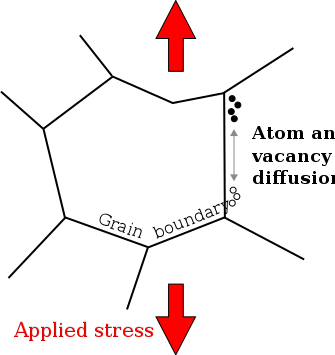
In materials science, Coble creep, a form of diffusion creep, is a mechanism for deformation of crystalline solids. Contrasted with other diffusional creep mechanisms, Coble creep is similar to Nabarro–Herring creep in that it is dominant at lower stress levels and higher temperatures than creep mechanisms utilizing dislocation glide.[1] Coble creep occurs through the diffusion of atoms in a material along grain boundaries. This mechanism is observed in polycrystals or along the surface in a single crystal, which produces a net flow of material and a sliding of the grain boundaries.
It was introduced by American materials scientist Robert L. Coble in 1962. Coble first reported his theory of how materials creep across grain boundaries and at high temperatures in alumina. Here he famously noticed a different creep mechanism that was more dependent on the size of the grain.[2]
The strain rate in a material experiencing Coble creep is given by where
- is a geometric prefactor
- is the applied stress,
- is the average grain diameter,
- is the grain boundary width,
- is the diffusion coefficient in the grain boundary,
- is the vacancy formation energy,
- is the activation energy for diffusion along the grain boundary
- is the Boltzmann constant,
- is the thermodynamic temperature (in kelvins)
- is the atomic volume for the material.
Coble creep, a diffusive mechanism, is driven by a vacancy (or mass) concentration gradient. The change in vacancy concentration from its equilibrium value is given by
This can be seen by noting that and taking a high temperature expansion, where the first term on the right hand side is the vacancy concentration from tensile stress and the second term is the concentration due to compressive stress. This change in concentration occurs perpendicular to the applied stress axis, while parallel to the stress there is no change in vacancy concentration (due to the resolved stress and work being zero).[2]
We continue by assuming a spherical grain, to be consistent with the derivation for Nabarro–Herring creep; however, we will absorb geometric constants into a proportionality constant . If we consider the vacancy concentration across the grain under an applied tensile stress, then we note that there is a larger vacancy concentration at the equator (perpendicular to the applied stress) than at the poles (parallel to the applied stress). Therefore, a vacancy flux exists between the poles and equator of the grain. The vacancy flux is given by Fick's first law at the boundary: the diffusion coefficient times the gradient of vacancy concentration. For the gradient, we take the average value given by where we've divided the total concentration difference by the arclength between equator and pole then multiply by the boundary width and length .
where is a proportionality constant. From here, we note that the volume change due to a flux of vacancies diffusing from a source of area is the vacancy flux times atomic volume :
where the second equality follows from the definition of strain rate: . From here we can read off the strain rate:
where has absorbed constants and the vacancy diffusivity through the grain boundary .
![{\displaystyle {\begin{aligned}{\frac {d\varepsilon _{C}}{dt}}\equiv {\dot {\varepsilon }}_{C}&=A_{C}{\frac {\delta '}{d^{3}}}{\frac {\sigma \Omega }{k_{\rm {B}}T}}D_{0}\exp \left(-{\frac {Q_{f}+Q_{m}}{k_{\rm {B}}T}}\right)\\[4pt]&=A_{C}{\frac {\delta '}{d^{3}}}{\frac {\sigma \Omega }{k_{\rm {B}}T}}D_{\rm {GB}}\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/2047df78864c0eb9a36ee0317ad5de89ca912088)




































