Copper(II) fluoride
Chemical compound
From Wikipedia, the free encyclopedia
Copper(II) fluoride or cupric fluoride is an inorganic compound with the chemical formula CuF2. The anhydrous form is a white, ionic, crystalline, hygroscopic salt with a distorted rutile-type crystal structure, similar to other fluorides of chemical formulae MF2 (where M is a metal). The dihydrate, CuF2·2H2O, is blue in colour.[6]

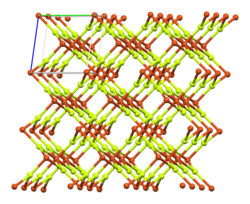
 Unit cell of the anhydrous form | |
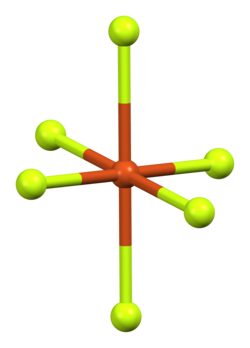
 Ball-and-stick model of crystal packing in the anhydrous form | |
 Dihydrate | |
| Names | |
|---|---|
| IUPAC name
Copper difluoride | |
| Other names
Cupric fluoride; Copper fluoride; Copper (2+) Difluoride | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.029.225 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| CuF2 | |
| Molar mass | 101.543 g/mol (anhydrous) 137.573 g/mol (dihydrate) |
| Appearance | White crystalline powder When hydrated: Blue |
| Density | 4.23 g/cm3 (anhydrous) 2.934 g/cm3 (dihydrate)[1] |
| Melting point | 836 °C (1,537 °F; 1,109 K) (anhydrous) 130 °C (dihydrate, decomposes) |
| Boiling point | 1,676 °C (3,049 °F; 1,949 K) (anhydrous) |
| +1050.0·10−6 cm3/mol | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
−267 kJ/mol (−63.8 kcal/mol) (gas)[2] |
Enthalpy of fusion (ΔfH⦵fus) |
54 kJ/mol (13 kcal/mol)[2] |
Enthalpy of sublimation (ΔfHsublim) |
267 kJ/mol (63.9 kcal/mol)[3] |
| Hazards | |
| GHS labelling:[4] | |
  | |
| Danger | |
| H302, H314, H332 | |
| P260, P264, P270, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P363, P405, P501 | |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 1 mg/m3 (as Cu)[5] |
REL (Recommended) |
TWA 1 mg/m3 (as Cu)[5] |
IDLH (Immediate danger) |
TWA 100 mg/m3 (as Cu)[5] |
| Related compounds | |
Other anions |
Copper(II) bromide Copper(II) chloride |
Other cations |
Silver(II) fluoride Cobalt(II) fluoride |
Related compounds |
Copper(I) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Properties
Copper(II) fluoride is slightly soluble in water, but starts to decompose in hot water, producing basic F− and Cu(OH)+ ions.[citation needed]
Structure
Copper(II) fluoride has a monoclinic crystal structure[7] and cannot achieve a higher-symmetry structure. It forms rectangular prisms with a parallelogram base.[8] Each copper ion has four neighbouring fluoride ions at 1.93 Å separation and two further away at 2.27 Å.[6] This distorted octahedral [4+2] coordination is a consequence of the Jahn–Teller effect in d9 copper(II),[9] and leads to a distorted rutile structure similar to that of chromium(II) fluoride, CrF2, which is a d4 compound.[6]
Synthesis
Uses
Cupric fluoride catalyzes the decomposition of nitric oxides in emission control systems.[11]
Copper(II) fluoride can be used to make fluorinated aromatic hydrocarbons by reacting with aromatic hydrocarbons in an oxygen-containing atmosphere at temperatures above 450 °C (842 °F). This reaction is simpler than the Sandmeyer reaction, but is only effective in making compounds that can survive at the temperature used. A coupled reaction using oxygen and 2 HF regenerates the copper(II) fluoride, producing water.[12] This method has been proposed as a "greener" method of producing fluoroaromatics since it avoids producing toxic waste products such as ammonium fluoride.
Reactions
It loses fluorine in the molten stage at temperatures above 950 °C (1742 °F).[citation needed]
- 2 CuF2 → 2 CuF + F2
- 2 CuF → CuF2 + Cu
The complex anions CuF3−, CuF42− and CuF64− are formed if CuF2 is exposed to substances containing fluoride ions F−.[citation needed]