Cubane
Organic compound (C8H8) with a cube carbon structure
From Wikipedia, the free encyclopedia
Cubane is a synthetic hydrocarbon compound with the formula C8H8. It consists of eight carbon atoms arranged at the corners of a cube, with one hydrogen atom attached to each carbon atom. A solid crystalline substance, cubane is one of the Platonic hydrocarbons and a member of the prismanes. It was first synthesized in 1964 by Philip Eaton and Thomas Cole.[4] Before this work, Eaton believed that cubane would be impossible to synthesize due to the "required 90 degree bond angles".[5][6] The cubic shape requires the carbon atoms to adopt an unusually sharp 90° bonding angle, which would be highly strained as compared to the 109.45° angle of a tetrahedral carbon. Once formed, cubane is quite kinetically stable, due to a lack of readily available decomposition paths. It is the simplest hydrocarbon with octahedral symmetry.

 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Cubane[1] | |||
| Systematic IUPAC name
Pentacyclo[4.2.0.02,5.03,8.04,7]octane | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C8H8 | |||
| Molar mass | 104.15 g/mol | ||
| Appearance | Transparent[2] crystalline solid | ||
| Density | 1.29 g/cm3 | ||
| Melting point | 133.5 °C (272.3 °F; 406.6 K)[3] | ||
| Boiling point | 161.6 °C (322.9 °F; 434.8 K)[3] | ||
| Related compounds | |||
Related hydrocarbons |
Cuneane Dodecahedrane Tetrahedrane Prismane Prismane C8 | ||
Related compounds |
Octafluorocubane Octanitrocubane Octaazacubane Mirex | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Having high potential energy and kinetic stability makes cubane and its derivative compounds useful for controlled energy storage. For example, octanitrocubane and heptanitrocubane have been studied as high-performance explosives. These compounds also typically have a very high density for hydrocarbon molecules. The resulting high energy density means a large amount of energy can be stored in a comparably smaller amount of space, an important consideration for applications in fuel storage and energy transport. Furthermore, their geometry and stability make them suitable isosteres for benzene rings.[7]
Synthesis
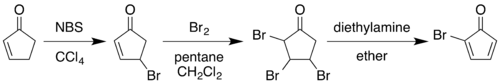
The classic 1964 synthesis starts with the conversion of 2-cyclopentenone to 2-bromocyclopentadienone:[4][8]
Allylic bromination with N-bromosuccinimide in carbon tetrachloride followed by addition of molecular bromine to the alkene gives a 2,3,4-tribromocyclopentanone. Treating this compound with diethylamine in diethyl ether causes elimination of two equivalents of hydrogen bromide to give the diene product.
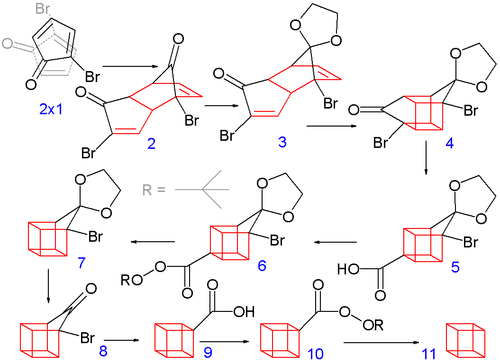
The construction of the eight-carbon cubane framework begins when 2-bromocyclopentadienone undergoes a spontaneous Diels-Alder dimerization. One ketal of the endo isomer is subsequently selectively deprotected with aqueous hydrochloric acid to 3.
In the next step, the endo isomer 3 (with both alkene groups in close proximity) forms the cage-like isomer 4 in a photochemical [2+2] cycloaddition. The bromoketone group is converted to ring-contracted carboxylic acid 5 in a Favorskii rearrangement with potassium hydroxide. Next, the thermal decarboxylation takes place through the acid chloride (with thionyl chloride) and the tert-butyl perester 6 (with tert-butyl hydroperoxide and pyridine) to 7; afterward, the acetal is once more removed in 8. A second Favorskii rearrangement gives 9, and finally another decarboxylation gives, via 10, cubane (11).
A more approachable laboratory synthesis of disubstituted cubane involves bromination of the ethylene ketal of cyclopentanone to give a tribromocyclopentanone derivative. Subsequent steps involve dehydrobromination, Diels-Alder dimerization, etc.[9][10]

The resulting cubane-1,4-dicarboxylic acid is used to synthesize other substituted cubanes. Cubane itself can be obtained nearly quantitatively by photochemical decarboxylation of the thiohydroxamate ester (the Barton decarboxylation).[11]
Reactions and derivatives

Cubane is highly strained, but cannot decompose because the resultant cubene molecules are pyramidal alkenes, too high-energy for most elimination pathways. Certain metallic ions catalyze σ-bond rearrangement to cuneane:[12][13]
With a rhodium catalyst, cubane first forms syn-tricyclooctadiene, which can thermally decompose to cyclooctatetraene at 50–60 °C.[14]
The main cubane functionalization challenge is C-H bond activation. Cubenes still inhibit decomposition during radical substitution, but the reaction offers little control against oversubstitution. In polar reactions, cubane reacts somewhat similarly to arenes or other cluster compounds: it metallates easily.[15] Cubane is slightly acidic, deprotonating about 63000 times faster than cyclohexane.[16]
Cubane substituents generally display normal reactivity. For example a Curtius rearrangement followed by organic oxidation converts cubane tetra(carbonylchloride) to tetranitrocubane.[15] However, electron-rich substituents such as alcohols can enable decomposition; they stabilize the cubene intermediate as a ketone (or equivalent) tautomer.[17]
Oxalyl chloride oxidizes cubane carboxamides to give a norcubane-furanone derivative.[18]
Hypercubane was predicted to exist in a 2014 publication.[19][20]
Persubstituted derivatives
Octaphenylcubane pre-dates the parent compound. Freedman synthesized it from tetraphenylcyclobutadiene nickel bromide in 1962. It is a sparingly soluble colourless compound that melts at 425–427 °C.[3][21][22][23]
Octanitrocubane is a green explosive.
Both heptafluorocubane and octafluorocubane were synthesized in 2022 to study octafluorocubane's unusual electronic structure.[24] Single-electron reduction to the radical anion C
8F−
8 traps[25] an otherwise-free electron inside the cube, making it the world's smallest box.[26]
Cubenes and oligo-cubylcubanes
Despite their orbital strain, two cubenes have been synthesized, and a third analyzed computationally. ortho-Cubene, produced via lithium-halogen exchange followed by elimination,[17] was the most pyramidalized alkene ever made at the time of its first synthesis.[27] In 2026, a new study of cubene was reported by Garg and Houk, showing that cubene has unusually low bond order and undergoes cycloadditions.[28]
Meta-cubene (1,3-dehydrocubane) is even less stable, and para-cubene (1,4-dehydrocubane) probably only exists as a diradical rather than an actual diagonal bond.[29] They rapidly undergo nucleophilic addition.[30]
Decomposition of cubenes has enabled chemists to synthesize cubylcubane, as well as higher oligomers.[30] Per X-ray diffraction, the central cubane-cubane bond is exceedingly short (1.458 Å), much shorter than the typical C-C single bond (1.578 Å). This is attributed to the fact that the exocyclic orbitals of cubane are s-rich and close to the nucleus.[31]
The oligo-cubylcubanes are rigid molecular rods considered for liquid crystal design. Absent solubilizing groups on the cubane monomer, oligomers with at least 4 units are essentially insoluble, thus scarcely accessible through conventional organic synthesis.[32]
poly-cubane

poly-cubane may be synthesized via solid-state polymerization of cubane at 250 °C and high pressure (4–30 GPa) in a diamond anvil cell, and is recoverable at ambient temperature and pressure.[33] It is a translucent yellow crystalline solid, denser than cubane itself, that is stable up to 300 °C.[33] Provisional structure assignment from powder X-ray diffraction suggests a model for the material that is formally derived from a 1D chain of cubane molecules by breaking two diametrically opposite C–C bonds in each cubane molecule to form a diasterane-based tetraradical, then connecting each such group to the groups directly to its left and right via pairs of C–C bonds.[33]: Figure 2 It exhibits an exceptionally high refractive index.[33][failed verification]