Decane
Alkane hydrocarbon; component of gasoline (petrol) and kerosene
From Wikipedia, the free encyclopedia
Decane is an alkane hydrocarbon with the chemical formula C10H22. Although 75 structural isomers are possible for decane, the term usually refers to the normal-decane ("n-decane"), with the formula CH3(CH2)8CH3. All isomers, however, exhibit similar properties and little attention is paid to the composition.[5] These isomers are flammable liquids. Decane is present in small quantities (less than 1%) in gasoline (petrol) and kerosene.[6][7] Like other alkanes, it is a nonpolar solvent, and does not dissolve in water, and is readily combustible. Although it is a component of fuels, it is of little importance as a chemical feedstock, unlike a handful of other alkanes.[8]
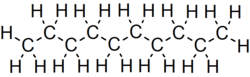 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Decane[1] | |
| Other names
Decyl hydride | |
| Identifiers | |
3D model (JSmol) |
|
| 1696981 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.004.262 |
| EC Number |
|
| MeSH | decane |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 2247 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H22 | |
| Molar mass | 142.286 g·mol−1 |
| Appearance | Colorless liquid |
| Odor | Gasoline-like (in high concentrations) |
| Density | 0.730 g mL−1 |
| Melting point | −30.5 to −29.2 °C; −22.8 to −20.6 °F; 242.7 to 243.9 K |
| Boiling point | 173.8 to 174.4 °C; 344.7 to 345.8 °F; 446.9 to 447.5 K |
| log P | 5.802 |
| Vapor pressure | 195 Pa[2] |
Henry's law constant (kH) |
2.1 nmol Pa−1 kg−1 |
| −119.74·10−6 cm3/mol | |
| Thermal conductivity | 0.1381 W m−1 K−1 (300 K)[3] |
Refractive index (nD) |
1.411–1.412 |
| Viscosity |
|
| Thermochemistry | |
Heat capacity (C) |
315.46 J K−1 mol−1 |
Std molar entropy (S⦵298) |
425.89 J K−1 mol−1 |
Std enthalpy of formation (ΔfH⦵298) |
−302.1 – −299.9 kJ mol−1 |
Std enthalpy of combustion (ΔcH⦵298) |
−6779.21 – −6777.45 kJ mol−1 |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Flammable, moderately toxic |
| GHS labelling: | |
  | |
| Danger | |
| H226, H302, H304, H305 | |
| P301+P310, P331 | |
| NFPA 704 (fire diamond) | |
| Flash point | 46.0 °C (114.8 °F; 319.1 K) |
| 210.0 °C (410.0 °F; 483.1 K) | |
| Explosive limits | 0.8–2.6% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
|
| Safety data sheet (SDS) | hazard.com |
| Related compounds | |
Related alkanes |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Reactions
Decane undergoes combustion, just like other alkanes. In the presence of sufficient oxygen, it burns to form water and carbon dioxide.
- 2 C10H22 + 31 O2 → 20 CO2 + 22 H2O
With insufficient oxygen, carbon monoxide is also formed.
It can be manufactured in the laboratory without fossil fuels.[9]
Physical properties
It has a surface tension of 0.0238 N·m−1.[10]

