Diethyl acetamidomalonate
From Wikipedia, the free encyclopedia
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.012.685 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H15NO5 | |
| Molar mass | 217.22 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
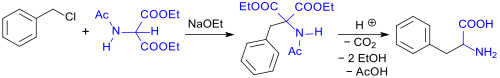
Diethyl acetamidomalonate (DEAM) is a derivative of malonic acid diethyl ester. Formally, it is derived through the acetylation of ester from the unstable aminomalonic acid. DEAM serves as a starting material for racemates including both, natural and unnatural α-amino acids or hydroxycarboxylic acids. It is also usable as a precursor in pharmaceutical formulations, particularly in the cases of active ingredients like fingolimod, which is used to treat multiple sclerosis.
A notable method for synthesizing acetamidomalon ester is described in a 1950 patent,[1] which cites a procedure previously featured in Organic Syntheses.[2] The synthesis procedure involves the preparation of malonic acid diethyl ester in acetic acid combined with sodium nitrite (NaNO2), resulting in diethyl isonitrosomalonate (also known as α-oximinomalonic acid diethyl ester).

The process further involves reducing a solution of this intermediate in a mixture of glacial acetic acid and acetic anhydride using zinc powder. The resulting amine is then combined with acetic anhydride to produce the final product DEAM, achieving a total yield of 77% acetylation.