Endocapillary proliferative glomerulonephritis
From Wikipedia, the free encyclopedia
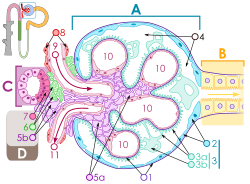
Endocapillary proliferative glomerulonephritis is a histological pattern with the basis of glomerular inflammation. It is characterized by endothelial hypercellularity (lining capillary loops) and white blood cell infiltrates resulting from subendothelial immune deposits. Both cells' distribution and expansion are restricted to the capillary lumen, within the glomerular basement membrane. Hence the histological pattern of endocapillary proliferative glomerulonephritis is marked by the presence of endomembranous deposits in the subendothelial space confined to the glomerular capillary filtration barrier.[1][2][3]
Immune antigen-antibody complexes
Different etiologies of proliferative GN share a commonality in almost all forms, the key pathological driver being the accumulation of immune deposits including antigen-antibody complexes, complement, and Ig[1] within the subendothelial space between the endothelium and glomerular basement membrane. This inciting trigger activates not only local inflammation, but also stimulates the complement system, resulting in an influx of inflammatory cells, including neutrophils and monocytes, alongside cell proliferation of endogenous endothelial cells. This intense hypercellularity and dense deposits cause capillary lumen occlusion and capillary wall thickening which compromises the filtration barrier, resulting in the leakage of proteins and RBCs in the urine (hematuria and proteinuria) alongside edema. These functional changes are congruent/correspondent to nephritic syndrome.
PIGN
- Post-streptococcal: PSGN: group A beta-hemolytic streptococcal infection results in the release of soluble cationic streptococcal antigens with high affinity for the negatively-charged GBM. These antigens form complexes either in-situ or in the peripheral blood. Once deposited the complexes cause type III HS activating the alternative complement pathway (reduces C3, normal C4) inducing leukocyte proliferation alongside endothelial and mesangial hypercellularity compromising glomerular filtration.
- Streptococcal antigens = M protein, nephritis-associated plasmin receptor, and streptococcal pyrogenic exotoxin B
- Post-staphylococcal: staphylococcal infections during active bacteremia produce super antigens that bind to MHC-II on antigen presenting cells, activating T-cells triggering dysregulated proliferation of T cells and B cells leading to a systemic cytokine storm. To manage bacteremia neutrophils create NETs that get trapped in glomerular capillaries causing endothelial damage.
- Staphylococcal super antigens = staphylococcal enterotoxins C, A, and toxic shock syndrome toxin-1
- Others: viral and parasitic (rare)[4]
IgA nephropathy
Faulty immunoglobulin causes dysregulated immune response triggered by previous infection, genetic (galactose-deficient IgA1), or idiopathic causes. Abnormal IgA are recognized as foreign, resulting in the production of anti-glycan autoantibodies. IgA complexes are circulating or formed in situ primarily in the mesangium. Deposits lead to lectin and alternative pathway complement activation causing an influx of macrophage and dendritic cells resulting in GBM damage and interstitial fibrosis.[5]
Lupus nephritis
Impaired apoptotic clearance of dead cells results in autoimmunity - nucleosome (DNA debris) lingers extracellularly causing anti-dsDNA autoantibodies [lupus autoimmunity] complexes may form in peripheral blood or deposit in-situ, initially confined to the mesangium.
Epitope spreading occurs with increase of complex burden, proportionally target proteins increase, expanding deposition to the subendothelium. Proliferation drives type III HS and triggers the classical complement cascade (low C3 and low C4) leading to leukocyte infiltration and kidney damage.[6]
- Class I/II = mesangial
- Class III/IV = sub endothelial space
Complement cascade
Pathogenesis of EPGN is fundamentally driven by the activation of the complement cascade. In autoimmune etiologies the classical pathway typically plays the major role, whereas other EPGN forms demonstrate a marked activation of the alternative pathway. This differential triggering translates to a distinct consumption pattern evident in the serum profile, resulting in significant C3 depletion (hypocomplementemia) while C4 levels remain relatively preserved.[7]
The consequent generation of potent chemotactic factors (C3a and C5a) provides the stimulus for a massive influx of inflammatory infiltrates. Polymorphonuclear leukocytes and monocytes are recruited directly to the glomerular capillaries, further driving the endocapillary hypercellularity and exacerbating the local glomerular inflammation.[8]
Monoclonal immunoglobulin deposit
Proliferative glomerulonephritis with monoclonal immunoglobulin deposits is a monoclonal gammopathy distinguished by a glomerular deposition of immunoglobulin causing kidney injury. It is a rare type of monoclonal gammopathy of renal significance. The glomerular injury occurs via a direct deposition mechanism of monoclonal immunoglobulin and leads to proliferative glomerulonephritis. The predominant immunoglobulin found in PGNMID is IgG3. It is limited by the glomerular filtration membrane due to its high molecular weight, which makes it prone to deposit on the glomerular capillary wall. The physiopathological mechanism giving rise to glomerular monoclonal immunoglobulin deposit is scarcely described and remains unclear. The deposition of IgG3 initiates the classical complement pathway as it has the ability to self-aggregate through Fc-Fc fragments and a high affinity at binding C1q.[9]