Ethylene (data page)
From Wikipedia, the free encyclopedia
This page provides supplementary chemical data on ethylene.
| Structure and properties | |
|---|---|
| Index of refraction | ? |
| Dielectric constant | ? ε0 at ? °C |
| Bond strength | ? |
| Bond length | C-C 133pm C-H 108 pm |
| Bond angle | 121.7 |
| Magnetic susceptibility | ? |
Thermodynamic properties
| Phase behavior | |
|---|---|
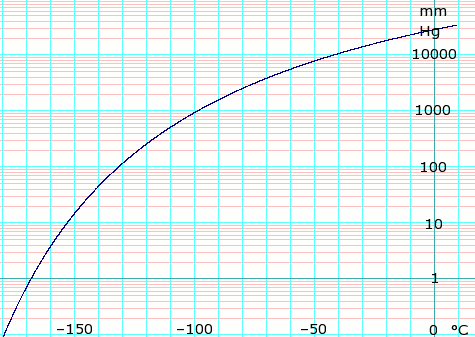
| Triple point | 104 K (−169 °C), 120 Pa |
| Critical point | 282.5 K (9.4 °C), 50.6 bar |
| Std enthalpy change of fusion, ΔfusH |
+3.35 kJ/mol |
| Std entropy change of fusion, ΔfusS |
+32.2 J/(mol·K) |
| Std enthalpy change of vaporization, ΔvapH |
+13.5 kJ/mol |
| Std entropy change of vaporization, ΔvapS |
? J/(mol·K) |
| Solid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
? J/(mol K) |
| Heat capacity, cp | ? J/(mol K) |
| Liquid properties | |
| Std enthalpy change of formation, ΔfH |
? kJ/mol |
| Standard molar entropy, S |
117.8 J/(mol K) |
| Heat capacity, cp | 67.4 J/(mol K) |
| Gas properties | |
| Std enthalpy change of formation, ΔfH |
+52.47 kJ/mol |
| Standard molar entropy, S |
219.32 J/(mol K) |
| Enthalpy of combustion, ΔcH |
–1387.4 kJ/mol |
| Heat capacity, cp | 42.9 J/(mol K) |
| van der Waals' constants[1] | a = 453.02 L2 kPa/mol2 b = 0.05714 liter per mole |