Hydrohalite
From Wikipedia, the free encyclopedia
| Hydrohalite | |
|---|---|
| General | |
| Category | Halide mineral |
| Formula | NaCl·2H2O |
| IMA symbol | Hhl[1] |
| Strunz classification | 3.BA.05 |
| Dana classification | 9.1.2.1 |
| Crystal system | Monoclinic |
| Crystal class | Prismatic (2/m) (same H-M symbol) |
| Space group | P21/c |
| Identification | |
| Colour | Colourless or white |
| Diaphaneity | Transparent |
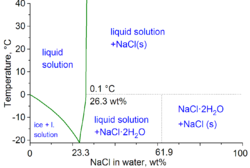
Hydrohalite is a halide mineral that occurs in saturated halite brines at cold temperatures (below 0.1 °C) and is the most common form of hydrated sodium chloride. It was first described in 1847 from an occurrence in Dürrnberg, Austria.