Indaziflam
From Wikipedia, the free encyclopedia
 | |
| Names | |
|---|---|
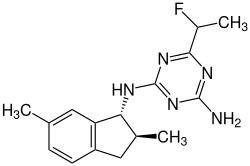
| IUPAC name
2-N-[(1R,2S)-2,6-dimethyl-2,3-dihydro-1H-inden-1-yl]-6-(1-fluoroethyl)-1,3,5-triazine-2,4-diamine[1] | |
| Identifiers | |
3D model (JSmol) |
|
| 20920435 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.216.692 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties[2] | |
| C16H20FN5 | |
| Molar mass | 301.369 g·mol−1 |
| Density | 1.23 g/mL |
| Melting point | 183 °C (361 °F; 456 K) |
| 2.8 mg/L (20 °C) | |
| log P | 2.8 |
| Hazards | |
| GHS labelling: | |
  | |
| Warning | |
| H373, H410 | |
| P260, P273, P314, P391, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Indaziflam is a preemergent herbicide used especially for grass control in tree and bush crops.
In 1991, the Japanese company Idemitsu Kosan filed a patent to 2-amino 6-fluoroalkyl triazine derivatives as herbicides.[3] One of these compounds was subsequently given the ISO common name triaziflam but had limited success as a commercial herbicide.[4][5] Bayer scientists subsequently investigated this area of chemistry and identified indaziflam as having superior properties, which they patented and developed under the code number BCS-AA10717.[6][7] The compound was first registered for use in the USA in 2010.[8][9]
Mechanism of action
Indaziflam is an inhibitor of cellulose biosynthesis. This mechanism of action was theorized to be responsible for indaziflam's effect in 2009[7] and proven in 2014.[10] The cellulose biosynthesis inhibitors (CBIs) are identified as Group 29 (Aus: Group O, WSSA: Group L) by the Weed Science Society of America/Herbicide Resistance Action Committee.[11][12]