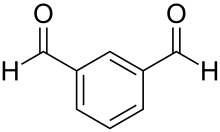
Isophthalaldehyde
From Wikipedia, the free encyclopedia
 | |
| Names | |
|---|---|
| Other names
1,3-Benzenedialdehyde, 1,3-Diformylbenzene | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.009.942 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H6O2 | |
| Molar mass | 134.134 g·mol−1 |
| Appearance | white |
| Density | 1.395 g/cm3[1] |
| Melting point | 89.5 °C (193.1 °F; 362.6 K) |
| Hazards | |
| GHS labelling:[2] | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Isophthalaldehyde is an organic compound with the formula C6H4(CHO)2. It is one of three isomers of benzene dicarbaldehyde, a reduced analog of phthalic acid. It is a colorless solid, although commercial samples often appear yellowish. One preparation entails the Sommelet reaction of α,α'-diamino-ortho-xylene.[3]