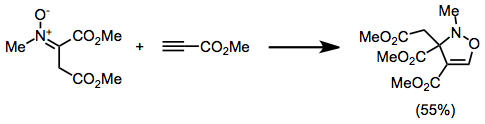Isoxazoline
From Wikipedia, the free encyclopedia
 2-isoxazoline | |
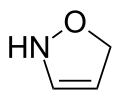 3-isoxazoline | |
 4-isoxazoline | |
| Names | |
|---|---|
| Systematic IUPAC name
Respective to images: 4,5-Dihydroisoxazole 2,5-Dihydroisoxazole 2,3-Dihydroisoxazole | |
| Other names
Respective to images: Δ2-isoxazoline Δ3-isoxazoline Δ4-isoxazoline | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H5NO | |
| Molar mass | 71.079 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Isoxazoline is a five-membered heterocyclic chemical compound, containing one atom each of oxygen and nitrogen which are located adjacent to one another. The ring was named in-line with the Hantzsch–Widman nomenclature. Isoxazolines are structural isomers of the more common oxazolines and exist in three different isomers depending on the location of the double bond. The relatively weak N-O bond makes isoxazolines prone to ring-opening and rearrangement reactions.
Isoxazoline insecticide
There are thousands of compounds containing isoxazoline rings, which are described to have a particular use. A number of naturally occurring isoxazolines with possible anti-cancer activity are produced by marine sponges.[1]
The isoxazoline class of insecticides was discovered by Nissan.[2] They act by allosterically modulating GABA-gated chloride channels (IRAC group 30) and glutamate-gated chloride channels; this decreases influx of chloride into the post-synaptic neuron, leading to hyperexcitation of the neuron, and paralysis of the parasite.[3]
Fluralaner and lotilaner are used against fleas in both dogs and cats, whereas afoxolaner and sarolaner are only approved for dogs,[3] and esafoxolaner is currently only approved for cats.[4] In 2018, the U.S. FDA issued a drug safety communication regarding post-marketing reports of neurological side effects in cats and dogs taking isoxazoline-containing flea & tick medications.[5][6]
Lotilaner is also the first isoxazoline approved for human use as Lotilaner eye drops, sold under the brand name Xdemvy. This medication is approved for treatment of blepharitis (inflammation of the eyelid) caused by infestation by Demodex (tiny mites).[7]
Fluxametamide and isocycloseram are used as agricultural insecticides.[8][9]