Lead stearate
From Wikipedia, the free encyclopedia
 | |
| Names | |
|---|---|
| Other names
Lead(2+) octadecanoate, lead(II) stearate, lead distearate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.012.733 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
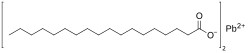
| C 36H 70PbO 4 | |
| Molar mass | 774.14 |
| Appearance | White powder |
| Density | 1.4 g/cm3 |
| Melting point | 115.7 °C (240.3 °F; 388.8 K) |
| Boiling point | 359.4 °C (678.9 °F; 632.5 K) |
| Slightly soluble | |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H302, H332, H360, H373 | |
| P260, P261, P281, P304, P340, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lead stearate is a metal-organic compound, a salt of lead and stearic acid with the chemical formula C
36H
70PbO
4.[1] The compound is classified as a metallic soap, i.e. a metal derivative of a fatty acid.[2] The compound is toxic.
The compound can be prepared by reacting stearic acid, lead(II) oxide, and a catalyst acetic acid.[3]
Also, an exchange reaction between lead(II) acetate and sodium stearate:

