Mabinlin
From Wikipedia, the free encyclopedia
| Mabinlin 1 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Organism | ? | ||||||
| Symbol | 2SS1_CAPMA | ||||||
| UniProt | P80351 | ||||||
| |||||||
| Mabinlin 2 | |||||||
|---|---|---|---|---|---|---|---|
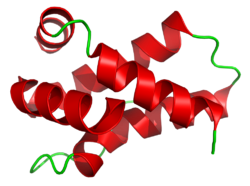 Also known as: Mabinlin II, MAB II | |||||||
| Identifiers | |||||||
| Organism | ? | ||||||
| Symbol | 2SS2_CAPMA | ||||||
| PDB | 2DS2 | ||||||
| UniProt | P30233 | ||||||
| |||||||
| Mabinlin 3 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Organism | ? | ||||||
| Symbol | 2SS3_CAPMA | ||||||
| UniProt | P80352 | ||||||
| |||||||
| Mabinlin 4 | |||||||
|---|---|---|---|---|---|---|---|
| Identifiers | |||||||
| Organism | ? | ||||||
| Symbol | 2SS4_CAPMA | ||||||
| UniProt | P80353 | ||||||
| |||||||

Mabinlins are sweet-tasting proteins extracted from the seed of mabinlang (Capparis masaikai Levl.), a plant growing in Yunnan province of China. There are four homologues. Mabinlin-2 was first isolated in 1983[1] and characterised in 1993,[2] and is the most extensively studied of the four. The other variants of mabinlin-1, -3 and -4 were discovered and characterised in 1994.[3]
The 4 mabinlins are very similar in their amino acids sequences (see below).
Chain A
M-1: EPLCRRQFQQ HQHLRACQRY IRRRAQRGGL VD
M-2: QLWRCQRQFL QHQRLRACQR FIHRRAQFGG QPD
M-3: EPLCRRQFQQ HQHLRACQRY LRRRAQRGGL AD
M-4: EPLCRRQFQQ HQHLRACQRY LRRRAQRG
Chain B
M-1: EQRGPALRLC CNQLRQVNKP CVCPVLRQAA HQQLYQGQIE GPRQVRQLFR AARNLPNICK IPAVGRCQFT RW
M-2: QPRRPALRQC CNQLRQVDRP CVCPVLRQAA QQVLQRQIIQ GPQQLRRLFD AARNLPNICN IPNIGACPFR AW
M-3: EQRGPALRLC CNQLRQVNKP CVCPVLRQAA HQQLYQGQIE GPRQVRRLFR AARNLPNICK IPAVGRCQFT RW
M-4: EQRGPALRLC CNQLRQVNKP CVCPVLRQAA HQQLYQGQIE GPRQVRRLFR AARNLPNICK IPAVGRCQFT RW
Amino acid sequence of Mabinlins homologues are adapted from Swiss-Prot biological database of protein.[4][5][6][7]
The molecular weights of Mabinlin-1, Mabinlin-3 and Mabinlin-4 are 12.3 kDa, 12.3 kDa and 11.9 kDa, respectively.[3]
With a molecular weight of 10.4kDa, mabinlin-2 is lighter than mabinlin-1. It is a heterodimer consisting of two different chains A and B produced by post-translational cleavage. The A chain is composed of 33 amino acid residues and the B chain is composed of 72 amino acid residues. The B chain contains two intramolecular disulfide bonds and is connected to the A chain through two intermolecular disulfide bridges.[2][8]
Mabinlin-2 is the sweet-tasting protein with the highest known thermostability,[9] which is due to the presence of the four disulfide bridges.[10] It has been suggested also that the difference in the heat stability of the different mabinlin homologues is due to the presence of an arginine residue (heat-stable homologue) or a glutamine (heat-unstable homologue) at position 47 in the B-chain.[3]
The B54-B64 segment of the beta chain in Mabinlin II is the primary functional domain that enables its function as a sweet protein.The segment contains the [NL/I] tetralit motif, a unique structural arrangement that contains four Asn-Leu/lle dipole units. The motif is stabilized by Pro56, Pro62, and a cysteine that is involved in a disulfide bridge, which includes CysB59-CysB11. The stabilization ensures a rigid and defined conformation.The region is one of the primary binding sites for receptor interaction such as the sweet receptor nTR2/T1R3.[11]
The sequences of Mabilins cluster with Napins (InterPro: IPR000617).
Sweetness properties
Mabinlins sweetness were estimated to be about 100–400 times that of sucrose on molar basis, 10 times sucrose on a weight basis,[2][3] which make them less sweet than thaumatin (3000 times) but elicit a similar sweetness profile.[12]
The sweetness of mabinlin-2 is unchanged after 48 hours incubation at 80 °C.[2]
Mabinlin-3 and -4 sweetness stayed unchanged after 1 hour at 80 °C, while mabinlin-1 loses sweetness after 1 hour at the same condition.[3][13]
As a sweetener
Mabinlins, as proteins, are readily soluble in water and found to be highly sweet; however, mabinlin-2 with its high heat stability has the best chance to be used as a sweetener.
During the past decade, attempts have been made to produce mabinlin-2 industrially. The sweet-tasting protein has been successfully synthesised by a stepwise solid-phase method in 1998, however the synthetic protein had an astringent-sweet taste.[8]
Mabinlin-2 has been expressed in transgenic potato tubers, but no explicit results have been reported yet.[14] However, patents to protect production of recombinant mabinlin by cloning and DNA sequencing have been issued.[15]