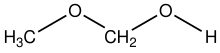
Methoxymethanol
From Wikipedia, the free encyclopedia
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Methoxymethanol | |
| Other names
Formaldehyde methyl hemiacetal | |
| Identifiers | |
3D model (JSmol) |
|
| 1900186 | |
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.022.476 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H6O2 | |
| Molar mass | 62.068 g·mol−1 |
| Density | 0.948 |
| Hazards | |
| GHS labelling: | |
   | |
| Warning | |
| H226, H302, H371 | |
| P210, P233, P240, P241, P242, P243, P260, P264, P270, P280, P301+P312, P303+P361+P353, P309+P311, P330, P370+P378, P403+P235, P405, P501 | |
| Flash point | 39.9 °C (103.8 °F; 313.0 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Methoxymethanol is a chemical compound which is both an ether and an alcohol, a hemiformal.[1] The structural formula can be written as CH3OCH2OH. It has been discovered in space.[2]