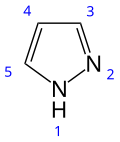
Pyrazole
Chemical compound
From Wikipedia, the free encyclopedia
Pyrazole is an organic compound with the formula (CH)3N2H. It is a heterocycle characterized as an azole with a 5-membered ring of three carbon atoms and two adjacent nitrogen atoms, which are in ortho-substitution. Pyrazoles are also a class of compounds that have the ring C3N2 with adjacent nitrogen atoms. Pyrazole itself has few applications but many substituted pyrazoles are of commercial interest. Notable drugs containing a pyrazole ring are celecoxib (celebrex) and the anabolic steroid stanozolol.
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1H-Pyrazole[1] | |||
| Systematic IUPAC name
1,2-Diazacyclopenta-2,4-diene | |||
| Other names
1,2-Diazole | |||
| Identifiers | |||
3D model (JSmol) |
|||
| 103775 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.005.471 | ||
| EC Number |
| ||
| 1360 | |||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C3H4N2 | |||
| Molar mass | 68.079 g·mol−1 | ||
| Melting point | 66 to 70 °C (151 to 158 °F; 339 to 343 K) | ||
| Boiling point | 186 to 188 °C (367 to 370 °F; 459 to 461 K) | ||
| Basicity (pKb) | 11.5 | ||
| Hazards | |||
| GHS labelling:[2] | |||
    | |||
| Danger | |||
| H302, H311, H315, H318, H319, H335, H372, H412 | |||
| P260, P262, P264, P264+P265, P270, P271, P273, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P305+P354+P338, P316, P317, P319, P321, P330, P332+P317, P337+P317, P361+P364, P362+P364, P403+P233, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Properties
Pyrazole is a weak base, with pKb 11.5 (pKa of the conjugate acid 2.49 at 25 °C).[3] According to X-ray crystallography, the compound is planar. The two C-N distances are similar, both near 1.33 Å[4]
History
The term pyrazole was given to this class of compounds by German Chemist Ludwig Knorr in 1883.[5] In a classical method developed by German chemist Hans von Pechmann in 1898, pyrazole was synthesized from acetylene and diazomethane.[6]
Preparation
Pyrazoles are synthesized by the reaction of α,β-unsaturated aldehydes with hydrazine and subsequent dehydrogenation:[7]
Substituted pyrazoles are prepared by condensation of 1,3-diketones with hydrazine (Knorr-type reactions).[8][9] For example, acetylacetone and hydrazine gives 3,5-dimethylpyrazole:[10]
- CH3C(O)CH2C(O)CH3 + N2H4 → (CH3)2C3HN2H + 2 H2O
A wide variety of pyrazoles can be made so:[8]
Occurrence and uses

In 1959, the first natural pyrazole, 1-pyrazolyl-alanine, was isolated from seeds of watermelons.[11][12]
In medicine, derivatives of pyrazole are widely used,[13] including celecoxib and similar COX-2 inhibitors, zaleplon, betazole, and CDPPB.[14] The pyrazole ring is found within a variety of pesticides as fungicides, insecticides and herbicides,[13] including fluoxapiprolin,[15] fenpyroximate, fipronil, tebufenpyrad and tolfenpyrad.[16] Pyrazole moieties are listed among the highly used ring systems for small molecule drugs by the US FDA[17]
3-(Difluoromethyl)-1-methyl-1H-pyrazole-4-carboxylic acid is used in the manufacture of six commercial fungicides which are inhibitors of succinate dehydrogenase.[18][19]
Pyrazole is an inhibitor of the alcohol dehydrogenase enzyme, and, as such, is used as an adjuvant with ethanol, to induce alcohol dependency in experimental laboratory mice.[20]
Conversion to scorpionates
Pyrazoles react with potassium borohydride to form a class of ligands known as scorpionate. Pyrazole itself reacts with potassium borohydride at high temperatures (~200 °C) to form a tridentate ligand known as Tp ligand:
See also
- 3,5-dimethylpyrazole
- Pyrazolidine, fully saturated analogue
- imidazole, structural analogue of pyrazole with two non-adjacent nitrogen atoms.
- isoxazole, another analogue, the nitrogen atom in position 1 replaced by oxygen.