Radical initiator
From Wikipedia, the free encyclopedia
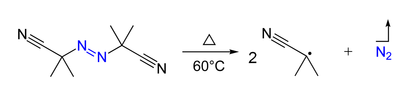
In chemistry, radical initiators are substances that can produce radical species under mild conditions and promote radical reactions.[1] These substances generally possess weak bonds—bonds that have small bond dissociation energies. Radical initiators are utilized in industrial processes such as polymer synthesis. Typical examples are molecules with a nitrogen-halogen bond, azo compounds, and organic and inorganic peroxides.[2]