Tetrabromoethylene
Chemical compound
From Wikipedia, the free encyclopedia
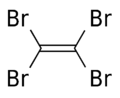
Tetrabromoethylene is an organobromine compound with the chemical formula C2Br4. Its structure is Br2C=CBr2. Under standard conditions, it exists in a form of colorless crystals. It is a brominated derivative of ethylene. Tetrabromoethylene is a potential fungicide and bactericide on fruits.[1] It was used in mineral separation.[2] It is an irritant.[3]
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Tetrabromoethene | |
| Other names
Perbromoethene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.001.084 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2Br4 | |
| Molar mass | 343.638 g·mol−1 |
| Appearance | Colorless crystals |
| Melting point | 50 °C (122 °F; 323 K) |
| Boiling point | 226 °C (439 °F; 499 K) |
| −114.8·10−6 cm3/mol | |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
It is prepared from acetylene and bromine in multiple steps.[1][4] One method involves dehydrobromination of pentabromoethane, other method involves bromination of dibromoethylene in chloroform.[1] Reaction of mercuric acetylide and bromine also gives tetrabromoethylene.[5] It can be produced by oxybrominating butane with free oxygen and bromine.[6]
Tetrabromoethylene gives tribromoacetyl bromide upon treatment with fuming nitric acid.[7]