Tetrachlorobenzene
Chemical compound
From Wikipedia, the free encyclopedia
Tetrachlorobenzene is any of three isomeric chlorobenzenes with the molecular formula C6H2Cl4. They differ by the positions of the chlorine atoms around the ring. Tetrachlorobenzenes are colorless crystalline compounds.[1]
 1,2,3,4-Tetrachlorobenzene | |
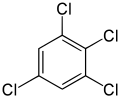 1,2,3,5-Tetrachlorobenzene | |
 1,2,4,5-Tetrachlorobenzene | |
| Identifiers | |
|---|---|
| |
3D model (JSmol) |
|
| ChEBI |
|
| ChEMBL |
|
| ChemSpider | |
| ECHA InfoCard | 100.032.390 |
| EC Number |
|
| KEGG |
|
PubChem CID |
|
| RTECS number |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H2Cl4 | |
| Molar mass | 215.88 g·molâ1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Properties
| isomer | m.p. (°C) | b.p. (°C) | m.p. (g/cm3 @100 °C) |
|---|---|---|---|
| 1,2,3,4 | 47 | 254 | 1.539 |
| 1,2,3,5 | 51.5 | 246 | 1.523 |
| 1,2,4,5 | 141 | 245 | 1.454 |
Synthesis
1,2,4,5-Tetrachlorobenzene can be produced by electrophilic halogenation of benzenes and some chlorobenzenes.[2] 1,2,3,4-Tetrachlorobenzene can only be produced by chlorination of 1,3,5-trichlorobenzene.
Uses
1,2,4,5-Tetrachlorobenzene once was used in the production of the herbicide 2,4,5-trichlorophenoxyacetic acid. This method has been discontinued because it produced highly toxic 2,3,7,8-tetrachlorodibenzo-p-dioxin as a waste product.[1][3][4]