1,2-Dioxolane
Chemical compound
From Wikipedia, the free encyclopedia
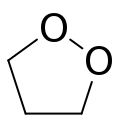
1,2-Dioxolane is a chemical compound with formula C3H6O2, consisting of a ring of three carbon atoms and two oxygen atoms in adjacent positions. Its condensed structural formula is [â(CH
2)3âOâOâ].[1][2]

 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,2-Dioxolane | |
| Systematic IUPAC name
1,2-Dioxacyclopentane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C3H6O2 | |
| Molar mass | 74.079 g·molâ1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The compound is an organic peroxide, specifically an endoperoxide, and a structural isomer of the much more common 1,3-dioxolane, which is often called simply "dioxolane".
Synthesis
Synthesis methods for the 1,2-dioxolane core structure include oxidation of cyclopropane derivatives with singlet oxygen[3] or molecular oxygen with a suitable catalyst,[4][5] the use of autooxidation, nucleophilic displacement with hydrogen peroxide, treatment with mercury(II) nitrate, photolysis of extended Ï-systems,[6] reaction of a bis-silylperoxide and an alkene,[7] or reaction with a 2-perhydroxy 4-alkene with diethylamine[8] or mercury(II) acetate.[9][10]
Occurrence
Some derivatives occur naturally, for example in Calophyllum dispar and from the seeds of the mamey (Mammea americana).[6] Plakinic acid A (3,5-peroxy 3Z,5Z,7,11-tetramethyl 13-phenyl-8E,12E-tridecadienoic acid) and similar compounds were isolated from sponges of the Plakortis genus.[11][12] Nardosinone is a sesquiterpene derivative with a 1,2-dioxolane element isolated from the plant Adenosma caeruleum.[13]
Uses
Synthetic and natural dioxolane derivatives have been used or considered as antimalarial drugs.[6][7] Plakinic acid A and related compounds showed antifungal action.[11]
