GHKL domain
From Wikipedia, the free encyclopedia
| Histidine kinase-, DNA gyrase B-, and HSP90-like ATPase | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
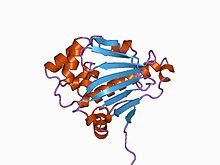 Structure of the N-terminal domain of the yeast Hsp90 chaperone.[1] | |||||||||||
| Identifiers | |||||||||||
| Symbol | HATPase_c | ||||||||||
| Pfam | PF02518 | ||||||||||
| InterPro | IPR003594 | ||||||||||
| SMART | HATPase_c | ||||||||||
| SCOP2 | 1ei1 / SCOPe / SUPFAM | ||||||||||
| CDD | cd00075 | ||||||||||
| |||||||||||
The GHKL domain (Gyrase, Hsp90, Histidine Kinase, MutL) is an evolutionary conserved protein domain.[2] It is an ATPase domain found in several ATP-binding proteins such as histidine kinase, DNA gyrase B, topoisomerases,[3] heat shock protein HSP90,[4][5][6] phytochrome-like ATPases and DNA mismatch repair proteins.
More information about this protein can be found at Protein of the Month: DNA Topoisomerase.[7]
The fold of this domain consists of two layers, alpha/beta, which contain an 8-stranded mixed beta-sheet.[2]