Ibogaline
From Wikipedia, the free encyclopedia
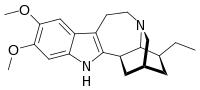 | |
| Names | |
|---|---|
| IUPAC name
(1R,17S)-17-Ethyl-6,7-dimethoxy-3,13-diazapentacyclo[13.3.1.02,10.04,9.013,18]nonadeca-2(10),4,6,8-tetraene | |
| Other names
12,13-Dimethoxyibogamine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C21H28N2O2 | |
| Molar mass | 340.467 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ibogaline is an alkaloid found in Tabernanthe iboga along with the related chemical compounds ibogaine, ibogamine, and other minor alkaloids. It is a relatively smaller component of Tabernanthe iboga root bark total alkaloids (TA) content. It is also present in Tabernaemontana species such as Tabernaemontana australis[1] which shares similar ibogan-biosynthetic pathways. The percentage of ibogaline in T. iboga root bark is up to 15% TA with ibogaine constituting 80% of the alkaloids and ibogamine up to 5%.[2][3]
Derivatives
Kisantine and Gabonine are thought to be ibogaline's oxidation byproducts.[4]