Phenylcopper
From Wikipedia, the free encyclopedia
 | |
| Names | |
|---|---|
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
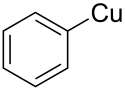
| C6H5Cu | |
| Molar mass | 140.652 g·mol−1 |
| Appearance | Colorless crystals |
| reacts with water | |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phenylcopper is an organometallic chemical compound of copper.[1] Its chemical formula is C6H5Cu,[2] where copper is in the oxidation state of +1.
Phenylcopper was the first known organocopper compound and was first prepared in 1923 from phenylmagnesium iodide and copper(I) iodide and in 1936 by Henry Gilman by transmetallation of phenylmagnesium iodide with copper(I) chloride.
Phenylcopper can be obtained by reacting phenyl lithium with copper(I) bromide in diethyl ether.[3]
- C6H5Li + CuBr → C6H5Cu + LiBr