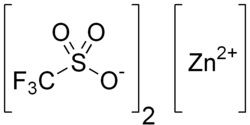
Zinc triflate
Chemical compound
From Wikipedia, the free encyclopedia
Zinc trifluoromethanesulfonate or zinc triflate is the zinc salt of trifluoromethanesulfonic acid. It is commonly used as a Lewis acid catalyst, e.g. in silylations.[1]

 | |
| Names | |
|---|---|
| IUPAC name
zinc trifluoromethanesulfonate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.053.548 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2F6O6S2Zn | |
| Molar mass | 363.51 g·mol−1 |
| Appearance | White powder |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
A white powder, zinc triflate is commercially available, though some workers have experienced inconsistent results with zinc triflate from different sources.[2][3] If desired, it may be prepared from reacting trifluoromethanesulfonic acid with zinc metal in acetonitrile,[4] or with zinc carbonate in methanol:[5]
- Zn + 2 HOTf → Zn(OTf)2 + H2
- ZnCO3 + 2 HOTf → Zn(OTf)2 + H2O + CO2 (OTf = CF3SO3)