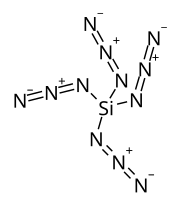
Silicon tetraazide
From Wikipedia, the free encyclopedia
| Names | |
|---|---|
| Other names
Tetraazidosilane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Si(N3)4 | |
| Molar mass | 196.1659 g/mol |
| Appearance | White crystals |
| Melting point | 212 °C (414 °F; 485 K) |
| Reacts | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Silicon tetraazide is a thermally unstable binary compound of silicon and nitrogen with a nitrogen content of 85.7% (by molar mass). This high-energy compound combusts spontaneously and can only be studied in a solution.[1][2][3] A further coordination to a six-fold coordinated structure such as a hexaazidosilicate ion [Si(N3)6]2−[4] or as an adduct with bidentate ligands Si(N3)4·L2[2] will result in relatively stable, crystalline solids that can be handled at room temperature.
Silicon tetraazide is synthesized by conversion of silicon tetrachloride with sodium azide in benzene.[1][3]
The reaction of silicon tetrachloride with an excess of sodium azide at room temperature in acetonitrile will result in the formation of sodium hexaazidosilicate (Na2[Si(N3)6]) which by adding ligands such as 2,2′-bipyridine and 1,10-phenanthroline will result in stable silicon tetraazide adducts.[2] Other bases such as pyridine and tetramethylethylenediamine will not react with the hexaazidosilicate ion.[2]
Another preparation of a bis(triphenylphosphine)iminium hexaazidosilicate salt [(Ph3P)2N]2[Si(N3)6] is possible by conversion of bis(triphenylphosphine)iminium azide [(Ph3P)2N]N3 with silicon tetrachloride in acetonitrile, where Ph is phenyl.[4]