2C (psychedelics)
Family of phenethylamine psychedelics
From Wikipedia, the free encyclopedia
2C (2C-x) is a general name for the family of psychedelic phenethylamines containing methoxy groups on the 2 and 5 positions of a benzene ring.[1][2][3] Most of these compounds also carry lipophilic substituents at the 4 position, usually resulting in more potent and more metabolically stable and longer acting compounds.[4]

Most of the early 2C drugs were developed by Alexander Shulgin in the 1970s and 1980s and were reviewed in his 1991 book PiHKAL (Phenethylamines I Have Known And Loved).[3][5][6] 2C-B is the most popular of the 2C drugs.[3]
Use and effects
The 2C drugs are orally active, are used at oral doses of 6 to 150 mg depending on the drug, and have durations of 3 to 48 hours depending on the drug.[1][7][6][8] However, many have doses in the range of 10 to 60 mg and durations in the range of 4 to 12 hours.[1] The 2C drugs produce psychedelic effects, such as perceptual enhancement, psychedelic visuals, and euphoria.[1][6][9][3] Some, such as 2C-B, have also been reported to produce some entactogen-like effects, but findings in this area appear to be mixed.[9][3][10][11]
| Compound | Chemical name | Dose | Duration | |
|---|---|---|---|---|
| 2C-AL | 4-Allyl-2,5-dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-B | 4-Bromo-2,5-dimethoxyphenethylamine | 10–35 mg | 4–8 hours | |
| 2C-Bu | 4-Butyl-2,5-dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-C | 4-Chloro-2,5-dimethoxyphenethylamine | 20–40 mg | 4–8 hours | |
| 2C-CN | 4-Cyano-2,5-dimethoxyphenethylamine | >22 mg | Unknown | |
| 2C-CP | 4-Cyclopropyl-2,5-dimethoxyphenethylamine | 15–35 mg | 3–6 hours | |
| 2C-D (2C-M) | 4-Methyl-2,5-dimethoxyphenethylamine | 20–60 mg | 4–6 hours | |
| 2C-E | 4-Ethyl-2,5-dimethoxyphenethylamine | 10–25 mg | 6–12 hours | |
| 2C-EF | 4-Fluoroethyl-2,5-dimethoxyphenethylamine | 10–25 mg | Unknown | |
| 2C-F | 4-Fluoro-2,5-dimethoxyphenethylamine | ≥250 mg | Unknown | |
| 2C-G (2C-G-0) | 3,4-Dimethyl-2,5-dimethoxyphenethylamine | 20–35 mg | 18–30 hours | |
| 2C-G-3 | 3,4-Trimethylene-2,5-dimethoxyphenethylamine | 16–25 mg | 12–24 hours | |
| 2C-G-5 | 3,4-Norbornyl-2,5-dimethoxyphenethylamine | 10–16 mg | 32–48 hours | |
| 2C-G-N | 1,4-Dimethoxynaphthyl-2-ethylamine | 20–40 mg | 20–30 hours | |
| 2C-H (2,5-DMPEA) | 2,5-Dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-I | 4-Iodo-2,5-dimethoxyphenethylamine | 14–22 mg | 6–10 hours | |
| 2C-iBu | 4-Isobutyl-2,5-dimethoxyphenethylamine | ≥5 mg | ~20 hours | |
| 2C-iP | 4-Isopropyl-2,5-dimethoxyphenethylamine | 8–25 mg | 8–12 hours | |
| 2C-N | 4-Nitro-2,5-dimethoxyphenethylamine | 100–150 mg | 4–6 hours | |
| 2C-O (2,4,5-TMPEA) | 4-Methoxy-2,5-dimethoxyphenethylamine | >300 mg | Unknown | |
| 2C-O-4 | 4-Isopropoxy-2,5-dimethoxyphenethylamine | >60 mg | Unknown | |
| 2C-O-22 | 4-(2,2,2-Trifluoroethoxy)-2,5-dimethoxyphenethylamine | ≥57 mg | Unknown | |
| 2C-P | 4-Propyl-2,5-dimethoxyphenethylamine | 6–10 mg | 5–16 hours | |
| 2C-Ph (2C-BI-1) | 4-Phenyl-2,5-dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-Se | 4-Methylseleno-2,5-dimethoxyphenethylamine | ~100 mg | 6–8 hours | |
| 2C-T (2C-T-1) | 4-Methylthio-2,5-dimethoxyphenethylamine | 60–100 mg | 3–5 hours | |
| 2C-T-2 | 4-Ethylthio-2,5-dimethoxyphenethylamine | 12–25 mg | 6–8 hours | |
| 2C-T-3 (2C-T-20) | 4-Methallylthio-2,5-dimethoxyphenethylamine | 15–40 mg | 8–14 hours | |
| 2C-T-4 | 4-Isopropylthio-2,5-dimethoxyphenethylamine | 8–20 mg | 12–18 hours | |
| 2C-T-7 | 4-Propylthio-2,5-dimethoxyphenethylamine | 10–30 mg | 8–15 hours | |
| 2C-T-8 | 4-Cyclopropylmethylthio-2,5-dimethoxyphenethylamine | 30–50 mg | 10–15 hours | |
| 2C-T-9 | 4-tert-Butylthio-2,5-dimethoxyphenethylamine | 60–100 mg | 12–18 hours | |
| 2C-T-13 | 4-(2-Methoxyethylthio)-2,5-dimethoxyphenethylamine | 25–40 mg | 6–8 hours | |
| 2C-T-15 | 4-Cyclopropylthio-2,5-dimethoxyphenethylamine | >30 mg | Several hours | |
| 2C-T-16 | 4-Allylthio-2,5-dimethoxyphenethylamine | 10–25 mg | 4–6 hours | |
| 2C-T-17 | 4-sec-Butylthio-2,5-dimethoxyphenethylamine | 60–100 mg | 10–15 hours | |
| 2C-T-19 | 4-Butylthio-2,5-dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-T-21 | 4-(2-Fluoroethylthio)-2,5-dimethoxyphenethylamine | 8–20 mg | 7–10 hours | |
| 2C-T-21.5 | 4-(2,2-Difluoroethylthio)-2,5-dimethoxyphenethylamine | 12–30 mg | 8–14 hours | |
| 2C-T-22 | 4-(2,2,2-Trifluoroethylthio)-2,5-dimethoxyphenethylamine | >10 mg | ~6 hours | |
| 2C-T-25 | 4-Isobutylthio-2,5-dimethoxyphenethylamine | >30 mg | Unknown | |
| 2C-T-27 | 4-Benzylthio-2,5-dimethoxyphenethylamine | ≥80 mg | Unknown | |
| 2C-T-28 | 4-(3-Fluoropropylthio)-2,5-dimethoxyphenethylamine | 8–20 mg | 8–10 hours | |
| 2C-T-30 | 4-(4-Fluorobutylthio)-2,5-dimethoxyphenethylamine | >8 mg | Unknown | |
| 2C-T-33 | 4-(3-Methoxybenzylthio)-2,5-dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-T-36 (2C-T-TFM) | 4-Trifluoromethylthio-2,5-dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-tBu | 4-tert-Butyl-2,5-dimethoxyphenethylamine | >5–10 mg | Unknown | |
| 2C-Te | 4-Methyltelluro-2,5-dimethoxyphenethylamine | Unknown | Unknown | |
| 2C-TFE | 4-(2,2,2-Trifluoroethyl)-2,5-dimethoxyphenethylamine | 5–15 mg | 12–24 hours | |
| 2C-TFM | 4-Trifluoromethyl-2,5-dimethoxyphenethylamine | 3–6 mg | ≥5–10 hours | |
| 2C-V | 4-Ethenyl-2,5-dimethoxyphenethylamine | ~25 mg | ~5 hours | |
| 2C-YN | 4-Ethynyl-2,5-dimethoxyphenethylamine | ~50 mg | ~2 hours | |
| Refs: [1][7][3][8][6][2][12][13][14][4] | ||||
Interactions
The 2C drugs are metabolized by the monoamine oxidase (MAO) enzymes, including both MAO-A and MAO-B.[1][15] As a result, they may be potentiated by monoamine oxidase inhibitors (MAOIs), such as phenelzine, tranylcypromine, moclobemide, and selegiline.[1][15][16] This may lead to overdose and serious toxicity.[1][15][16][17] There are anecdotal reports of strong potentiation of 2C-B by MAOIs, including hospitalization.[17] There is no known reversal agent for 2C drugs, and medical management for overdose involves treatment of symptoms until toxicity within the body subsides.[1]
Pharmacology
Pharmacodynamics
Actions
The 2C drugs act as agonists of the serotonin 5-HT2 receptors, including of the serotonin 5-HT2A, 5-HT2B, and 5-HT2C receptors.[18][19][20][21][22] They are partial agonists of the serotonin 5-HT2A receptor.[18][19] Most of the 2C drugs have much lower affinity for the serotonin 5-HT1A receptor than for the serotonin 5-HT2A receptor.[18][19][20][21] Most of the 2C drugs have also shown about 5- to 15-fold higher affinity for the serotonin 5-HT2A receptor over the serotonin 5-HT2C receptor and about 15- to 100-fold higher affinity for the serotonin 5-HT2A receptor over the serotonin 5-HT1A receptor.[19] The psychedelic effects of the 2C drugs are thought to be mediated specifically by activation of the serotonin 5-HT2A receptor.[18][20][22]
Unlike many other phenethylamines, 2C drugs, including 2C-C, 2C-D, 2C-E, 2C-I, and 2C-T-2 among others, are inactive as monoamine releasing agents and reuptake inhibitors.[18][23][20][19][22] Most of the 2C drugs are agonists of the rat and mouse trace amine-associated receptor 1 (TAAR1).[18][24][25][19] However, most are inactive as agonists of the human TAAR1.[18][24][25][19] The 2C drugs show very weak monoamine oxidase inhibition, including of monoamine oxidase A (MAO-A) and/or monoamine oxidase B (MAO-B).[18]
| Drug | 5-HT1A | 5-HT1B | 5-HT2A | 5-HT2B | 5-HT2C | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ki (nM) | EC50 (nM) | Emax (%) | Ki (nM) | Ki (nM) | EC50 (nM) | Emax (%) | Ki (nM) | EC50 (nM) | Emax (%) | Ki (nM) | EC50 (nM) | Emax (%) | |
| 2C-B | 130–311 | ND | ND | 104.4 | 6.9–27.6 | 1.89–80 | 5–99% | 13.5 | 75–130 | 52–89% | 43–89.5 | 0.031–0.264 | 104–116% |
| 2C-C | 190–740 | >10,000 | <25% | 252.9 | 5.47–13 | 9.27–200 | 49–102% | ND | 280 | 81% | 5.4–90 | 24.2 | 94% |
| 2C-D | 440–1,630 | >10,000 | <25% | ND | 23.9–32.4 | 43.5–350 | 41–125% | ND | 230 | 77% | 12.7–150 | 71.1 | 100% |
| 2C-E | 307.3–1,190 | >10,000 | <25% | ND | 4.50–43.9 | 2.5–110 | 40–125% | 25.1 | 190 | 66% | 5.4–104.1 | 0.233–18.0 | 98–106% |
| 2C-H | 70 | ND | ND | ND | 1,600 | 2,408–9,400 | 28–67% | ND | 6,200 | 46% | 4,100 | ND | ND |
| 2C-I | 180–970 | 4,900 | 102% | ND | 3.5–9.3 | 3.83–60 | 15–82% | ND | 150 | 70% | 10.2–40 | 2.8 | 79–100% |
| 2C-N | 2,200 | ND | ND | ND | 23.5 | 170 | 20–48% | ND | 730 | 74% | 370 | ND | 40–50% |
| 2C-P | 110 | ND | ND | ND | 8.1 | 90 | 63% | ND | 130 | 72% | 40 | ND | ND |
| 2C-T-1 | 1,035 | ND | ND | ND | 49 | 2.0 | 75% | ND | 57 | 58% | 347 | ND | ND |
| 2C-T-2 | 370–1,740 | 3,000 | 76% | 857.5 | 9–39.9 | 0.354–80 | 67–128% | 6 | 130 | 75% | 14.2–69 | 0.0233–3.8 | 87–107% |
| 2C-T-4 | 470–916 | ND | ND | ND | 27.9–54 | 5.5–220 | 56–87% | ND | 63–160 | 68–75% | 180–295 | ND | ND |
| 2C-T-7 | 520–878 | ND | ND | ND | 5.3–6.5 | 1.2–130 | 49–101% | ND | 52–350 | 45–75% | 39–54 | ND | ND |
| Notes: The smaller the value, the more avidly the drug binds to or activates the site. Refs: [19][20][21][18][26][27][28][29] | |||||||||||||
Effects
In accordance with their psychedelic effects in humans, the 2C drugs produce the head-twitch response and wet dog shakes, behavioral proxies of psychedelic effects, in rodents.[18] At least some 2C drugs, such as 2C-D and 2C-E, produce hyperlocomotion at lower doses in rodents.[18] All 2C drugs produce hypolocomotion at higher doses in rodents.[18] 2C drugs, including 2C-C, 2C-D, 2C-E, and 2C-I, substitute partially to fully for psychedelics like DOM, DMT, and LSD and/or for the entactogen MDMA in rodent drug discrimination tests.[18][20] However, none of the assessed 2C drugs substituted for dextromethamphetamine, suggesting that they lack amphetamine-type or stimulant-like effects.[18][20]
In contrast to most psychedelics, at least two assessed 2C drugs, 2C-C and 2C-P, have shown reinforcing effects in rodents, including conditioned place preference (CPP) and self-administration.[18][30] The mechanism by which these effects are mediated is unknown.[18] However, it may be related to reduced expression of the dopamine transporter (DAT) and increased DAT phosphorylation, in turn resulting in increased extracellular dopamine levels in certain brain areas.[18][30] These 2C drugs might have misuse potential in humans.[18][30] Similar reinforcing effects in animals have been observed for NBOMe analogues of 2C drugs, including 25B-NBOMe, 25D-NBOMe, 25E-NBOMe, 25H-NBOMe, and 25N-NBOMe.[18][31][32][33][34][35][36]
Similarly to DOI, tolerance has been found to gradually develop to the head-twitch response induced by 2C-T-7 with chronic administration in rodents.[18]
Various 2C drugs show potent anti-inflammatory effects mediated by serotonin 5-HT2A receptor activation.[37] Among these include 2C-I, 2C-B, 2C-H, and 2C-iBu.[37][38] Others, such as 2C-B-Fly and 2C-T-33, were less effective.[37] 2C-iBu has shown a greater separation between anti-inflammatory effects and psychedelic-like effects in animals than other 2C drugs and is being investigated for possible use as a pharmaceutical drug.[38][39]
Pharmacokinetics
The 2C drugs are orally active.[1] They are metabolized by O-demethylation and deamination.[1][15] This is mediated specifically by monoamine oxidase (MAO) enzymes MAO-A and MAO-B, whereas cytochrome P450 enzymes appear to metabolize only some 2C drugs and to have only a very small role.[15]
Chemistry
The 2C drugs, also known as 4-substituted 2,5-dimethoxyphenethylamines, are substituted phenethylamines and can be thought of as synthetic analogues of the naturally occurring phenethylamine psychedelic mescaline (3,4,5-trimethoxyphenethylamine).[5][6][7][40][4] They are the phenethylamine (2C) analogues of the amphetamine (α-methylphenethylamine) DOx drugs like DOM, DOB, and DOI as well as of the phenylisobutylamine (α-ethylphenethylamine) 4C drugs like Ariadne (4C-D) and 4C-B.[6][7][40][4] The N-benzylphenethylamines such as 25I-NBOMe, 25B-NBOMe, and 25C-NBOMe are derivatives of the 2C drugs.[41][5][4] Certain FLY drugs such as 2C-B-FLY are also 2C derivatives.[5][4][2]
Syntheses
The chemical syntheses of 2C drugs have been described.[6][2]
Analysis
The chemical analysis of 2C drugs has been described.[5]
History
2,4,5-Trimethoxyphenethylamine (2,4,5-TMPEA; 2C-O), the 2C positional isomer of mescaline (3,4,5-trimethoxyphenethylamine), was first synthesized by Max Jansen and was reported to produce psychedelic effects similar to those of mescaline in 1931.[42][43] However, subsequent studies in the 1960s and 1970s suggested that 2,4,5-TMPEA may actually be inactive as a psychedelic in animals and humans.[42]
2C-D was the first of the 2C drugs after 2C-O to be discovered.[2][44][45][46] It was synthesized and studied in animals by Beng T. Ho and colleagues at the Texas Research Institute of Mental Sciences and they published their findings in 1970.[2][44][45][46] Alexander Shulgin synthesized 2C-B and 2C-D in 1974 and discovered their psychedelic effects in self-experiments conducted in 1974 and 1975.[1][41][2][44][47][48] He published his findings in the scientific literature in 1975.[1][41][2][44][47] However, Shulgin had previously tested sub-threshold doses of 2C-D in 1964 and 1965.[49] 2C-T was first described by Shulgin and David E. Nichols in 1976.[50] 2C-I was first described by Shulgin and colleagues in 1977 and initial psychoactivity was reported by Shulgin in 1978.[42][51] Shulgin also first synthesized 2C-E in 1977.[52][53] He reviewed several of these 2C drugs in a literature review in 1979.[54] Subsequently, numerous other 2C drugs have been synthesized and characterized.[6][7][2][1][41] Shulgin comprehensively reviewed and described the 2C drugs in his 1991 book PiHKAL (Phenethylamines I Have Known and Loved).[6][3] He coined the term "2C", this term being an acronym for the two carbon atoms between the benzene ring and the amino group of the 2C drugs and a means to distinguish them from the three-carbon DOx drugs.[6][1][3]
2C-D was extensively studied by Hanscarl Leuner under the names DMM-PEA and LE-25 in psychedelic-assisted psychotherapy in Germany in the 1970s and 1980s.[41][55][56][57][58] It was also informally studied by Darrell Lemaire as a potential "smart drug" in the 1970s and 1980s.[59][60][61][62] He additionally developed the TWEETIO drugs such as 2CD-5-ETO via structural modification of the 2Cs.[59][60][61][62][40][7] 2C-B was legitimately marketed and sold as an over-the-counter sexual enhancer under brand names like Erox in several European countries such as Germany in the 1980s and early 1990s.[63][5][64][65] It was sold in adult stores, smart shops, and some nightclubs.[63][64]
2C-B was first encountered as a novel designer drug in the United States in 1979.[63] It gained popularity as a recreational drug and MDMA (ecstasy) alternative in the mid-1980s.[1][3][5] The drug became a controlled substance in the United States in 1994 or 1995.[1][3][5] It has been said to be the most popular of the 2C drugs in terms of recreational use.[3][5] Numerous other 2C drugs besides 2C-B have also since been made controlled substances.[5]
Society and culture
List of 2C drugs
| Name | R3 | R4 | Structure | CAS # |
|---|---|---|---|---|
| 2C-B | H | Br |  |
66142–81–2 |
| 2C-Bn | H | CH2C6H5 |  |
|
| 2C-Bu | H | CH2CH2CH2CH3 |  |
|
| 2C-C | H | Cl | 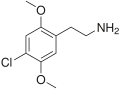 |
88441–14–9 |
| 2C-C-3 [68] | Cl | Cl |  |
|
| 2C-CN | H | C≡N |  |
88441–07–0 |
| 2C-D | H | CH3 |  |
24333–19–5 |
| 2C-E | H | CH2CH3 |  |
71539–34–9 |
| 2C-EF | H | CH2CH2F |  |
1222814–77–8 |
| 2C-F | H | F | 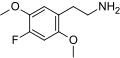 |
207740–15–6 |
| 2C-G (2C-G-0) | CH3 | CH3 | 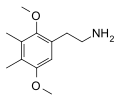 |
207740–18–9 |
| 2C-G-1 | CH2 |  |
2888537-47-9 | |
| 2C-G-2 | (CH2)2 |  |
2888537-48-0 | |
| 2C-G-3 | (CH2)3 |  |
207740–19–0 | |
| 2C-G-4 | (CH2)4 |  |
952006–59–6 | |
| 2C-G-5 | (CH2)5 |  |
207740–20–3 | |
| 2C-G-6 | (CH2)6 |  |
2888537-49-1 | |
| 2C-G-N | (CH)4 |  |
207740–21–4 | |
| 2C-H | H | H | 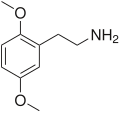 |
3600–86–0 |
| 2C-I | H | I |  |
69587–11–7 |
| 2C-iBu | H | iBu |  |
|
| 2C-iP | H | CH(CH3)2 |  |
1498978–47–4 |
| 2C-tBu | H | C(CH3)3 |  |
|
| 2C-CP | H | C3H5 |  |
2888537–46–8 |
| 2C-CB | H | C4H7 |  |
|
| 2C-CPe[69] | H | C5H9 |  |
|
| 2C-CPM | H | C4H7 |  |
|
| 2C-N | H | NO2 | 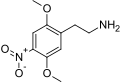 |
261789–00–8 |
| 2C-NH2 | H | NH2 |  |
168699–66–9 |
| 2C-PYR | H | Pyrrolidine |  |
|
| 2C-PIP [70] | H | Piperidine |  |
|
| 2C-O | H | OCH3 |  |
15394–83–9 |
| 2C-O-4 | H | OCH(CH3)2 |  |
952006–65–4 |
| 2C-O-22 | H | OCH2CF3 |  |
|
| 2C-MOM [71] | H | CH2OCH3 |  |
|
| 2C-P | H | CH2CH2CH3 |  |
207740–22–5 |
| 2C-Ph (2C-BI-1) | H | C6H5 |  |
|
| 2C-Se | H | SeCH3 |  |
1189246–68–1 |
| 2C-Se-TFM | H | SeCF3 |  |
? |
| 2C-Te | H | TeCH3 |  |
? |
| 2C-Te-TFM | H | TeCF3 |  |
? |
| 2C-T | H | SCH3 |  |
61638–09–3 |
| 2C-T-2 | H | SCH2CH3 | 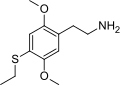 |
207740–24–7 |
| 2C-T-3[72] | H | SCH2C(=CH2)CH3 |  |
648957–40–8 |
| 2C-T-4 | H | SCH(CH3)2 | 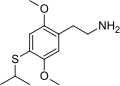 |
207740–25–8 |
| 2C-T-5[72] | H |  |
||
| 2C-T-6[72] | H | SC6H5 |  |
|
| 2C-T-7 | H | S(CH2)2CH3 |  |
207740–26–9 |
| 2C-T-8 | H | SCH2CH(CH2)2 | 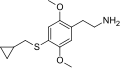 |
207740–27–0 |
| 2C-T-9[72] | H | SC(CH3)3 |  |
207740–28–1 |
| 2C-T-10[72] | H |  |
||
| 2C-T-11[72] | H | SC6H4-p-Br |  |
|
| 2C-T-12[72] | H |  |
||
| 2C-T-13 | H | S(CH2)2OCH3 |  |
207740–30–5 |
| 2C-T-14[72] | H | S(CH2)2SCH3 |  |
|
| 2C-T-15 | H | SCH(CH2)2 |  |
|
| 2C-T-16[73] | H | SCH2CH=CH2 |  |
648957–42–0 |
| 2C-T-17 | H | SCH(CH3)CH2CH3 | 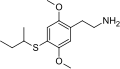 |
207740–32–7 |
| 2C-T-18[72] | H |  |
||
| 2C-T-19 | H | SCH2CH2CH2CH3 |  |
|
| 2C-T-21 | H | S(CH2)2F | 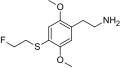 |
207740–33–8 |
| 2C-T-21.5[72] | H | S(CH2)CHF2 |  |
648957–46–4 |
| 2C-T-22[72] | H | S(CH2)CF3 |  |
648957–48–6 |
| 2C-T-23[72] | H | 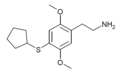 |
||
| 2C-T-24[72] | H |  |
||
| 2C-T-25[72] | H | SCH2CH(CH3)2 |  |
|
| 2C-T-27[72] | H | SCH2C6H5 |  |
648957–52–2 |
| 2C-T-28[72] | H | S(CH2)3F |  |
648957–54–4 |
| 2C-T-29 (2C-T-PARGY) | H | S(CH2)C≡CH |  |
|
| 2C-T-30[72] | H | S(CH2)4F |  |
|
| 2C-T-31[72] | H | SCH2C6H4-p-CF3 |  |
|
| 2C-T-32[72] | H | SCH2C6F5 |  |
|
| 2C-T-33[72] | H | SCH2C6H4-m-OCH3 |  |
|
| 2C-T-34 (2C-T-FM) | H | SCFH2 |  |
|
| 2C-T-35 (2C-T-DFM) | H | SCF2H |  |
|
| 2C-T-36 (2C-T-TFM; CYB210010)[74] | H | SCF3 |  |
|
| 2C-T-CH2CN | H | S(CH2)C≡N |  |
|
| 2C-T-pent-4-ynyl | H | S(CH2)3C≡CH |  |
|
| 2C-T-TFM-sulfone | H | SO2CF3 |  |
|
| 2C-T-DFP | H | SCH2CH2CF2H |  |
|
| 2C-T-TFP | H | SCH2CH2CF3 |  |
|
| 2C-DFM [4]: 770 | H | CHF2 |  |
|
| 2C-TFM | H | CF3 |  |
159277–08–4 |
| 2C-TFE | H | CH2CF3 |  |
|
| 2C-PFE | H | CF2CF3 | 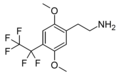 |
|
| 2C-PFS | H | SF5 |  |
|
| 2C-YN | H | C≡CH |  |
752982–24–4 |
| 2C-V | H | CH=CH2 |  |
|
| 2C-AL[75] | H | CH2CH=CH2 |  |
|
| 2C-1MV | H | 1-Methylvinyl |  |
|
| 2C-MAL | H | Methallyl |  |
|
Related compounds
| Name | Chemical name | Structure | Ref |
|---|---|---|---|
| 2C-DB (6-bromo-2C-B) | 2,5-Dimethoxy-4,6-dibromophenethylamine |  |
|
| N-Methyl-2C-B (2C-B-M, 2C-BM) | N-Methyl-4-bromo-2,5-dimethoxyphenethylamine |  |
|
| N-Ethyl-2C-B | N-Ethyl-4-bromo-2,5-dimethoxyphenethylamine |  |
|
| 2C-B-OH (N-hydroxy-2C-B) | 4-Bromo-2,5-dimethoxy-N-hydroxyphenethylamine |  |
[76] |
| 25B-NB (N-benzyl-2C-B) | N-Benzyl-4-bromo-2,5-dimethoxyphenethylamine |  |
|
| N-Methyl-2C-I | N-Methyl-4-iodo-2,5-dimethoxyphenethylamine |  |
|
| β-Methyl-2C-B (BMB) | 4-Bromo-2,5-dimethoxy-β-methylphenylethylamine |  |
|
| β-Keto-2C-B (βk-2C-B) | 4-Bromo-2,5-dimethoxy-β-ketophenylethylamine |  |
|
| β-Keto-2C-I (βk-2C-I) | 4-Iodo-2,5-dimethoxy-β-ketophenylethylamine |  |
|
| 2C-B-AN (2C-B-aminonitrile) | 4-Bromo-N-(α′-cyanobenzyl)-2,5-dimethoxyphenethylamine |  |
|
| 25D-NM-NDEAOP (25D-NM-NDEPA) | N-Methyl-N-(3-diethylamino-3-oxopropyl)-2,5-dimethoxy-4-methylphenethylamine |  |
|
| 25B-NAcPip | N-(Piperidin-1-ylcarbonylmethyl)-4-bromo-2,5-dimethoxyphenethylamine |  |
|
| XOB (ASR-6001) | N-[(4-Phenylbutoxy)hexyl]-4-bromo-2,5-dimethoxyphenethylamine | [77] | |
| TCB-2 (2CBCB, 2C-BCB) | [3-Bromo-2,5-dimethoxy-bicyclo[4.2.0]octa-1,3,5-trien-7-yl]methanamine |  |
|
| 2CB-Ind | (5-Bromo-4,7-dimethoxy-2,3-dihydro-1H-inden-1-yl)methanamine |  |
|
| ZC-B (2C-B-AZET) | 3-(4-Bromo-2,5-dimethoxyphenyl)azetidine |  |
|
| 2C-B-PYR | 3-(4-Bromo-2,5-dimethoxyphenyl)pyrrolidine |  |
|
| 2C-B-3PIP | 3-(4-Bromo-2,5-dimethoxyphenyl)piperidine |  |
|
| LPH-5 ((S)-2C-TFM-3PIP) | (S)-3-(2,5-Dimethoxy-4-(trifluoromethyl)phenyl)piperidine |  |
|
| DEMPDHPCA-2C-D ("compound 45") | 1-Methyl-3-(1-oxo-1-diethylaminomethyl)-5-(2,5-dimethoxy-4-methylphenyl)-3,6-dihydro-2H-pyridine |  |
[78] |
| DOM-CR (DOM-THIQ, 2C-D-CR) | 5,8-Dimethoxy-7-methyl-1,2,3,4-tetrahydroisoquinoline |  |
|
| DOB-CR (DOB-THIQ, 2C-B-CR) | 5,8-Dimethoxy-7-bromo-1,2,3,4-tetrahydroisoquinoline |  |
|
| N-Methyl-DOM-CR (Beatrice-CR, N-methyl-2C-D-CR) | 2,7-Dimethyl-5,8-dimethoxy-1,2,3,4-tetrahydroisoquinoline |  |
|
| 2C-B-morpholine (2C-B-MOR) | 2-(4-Bromo-2,5-dimethoxyphenyl)morpholine |  |
[79][80] |
| 2C-B-aminorex (2C-B-AR) | 5-(4-Bromo-2,5-dimethoxyphenyl)-4,5-dihydro-1,3-oxazol-2-amine |  |
|
| 2C-B-PP | 1-(2,5-Dimethoxy-4-bromophenyl)piperazine |  |
|
| 2C-B-BZP | 1-[(4-Bromo-2,5-dimethoxyphenyl)methyl]piperazine |  |
|
| 2C-B-5-hemiFLY-α6 (BNAP) | 8-Bromo-6-methoxy-2a,3,4,5-tetrahydro-2H-naphtho[1,8-bc]furan-4-amine |  |
|
| 2CB7 (2C-B-5-hemiFLY-β7) | (5-Bromo-7-methoxy-3-oxatricyclo[6.4.1.04,13]trideca-4,6,8(13)-trien-9-yl)methanamine |  |
|
| 2-OH-2C-B (2-DM-2C-B; B-2-HMPEA) | 4-Bromo-2-hydroxy-5-methoxyphenethylamine |  |
|
| 2C-2-TOM (2-thio-2C-D) | 5-Methoxy-4-methyl-2-methylthiophenethylamine |  |
[6][81] |
| 2C-5-TOM (5-thio-2C-D) | 2-Methoxy-4-methyl-5-methylthiophenethylamine |  |
[6][81] |
| 2C-2-TOET (2-thio-2C-E) | 4-Ethyl-5-methoxy-2-methylthiophenethylamine |  |
[6][81] |
| 2C-5-TOET (5-thio-2C-E) | 4-Ethyl-2-methoxy-5-methylthiophenethylamine |  |
[6][81] |
| 2T-2CTFM-3PIP (2-thio-LPH-5) | 3-(5-Methoxy-2-methylthio-4-(trifluoromethyl)phenyl)piperidine |  |
[82] |
